Fresh air on Mars: bend a CO2 molecule and get oxygen

What do we remember with the phrase "science fiction"? Of course, robots, flying cars, the study of previously unexplored corners of the Universe, aliens and a whole series of plots. Among them, a special place has always been occupied by the theme of settling on another planet, either from confidence in the inevitability of the death of one's own, or from the desire to radically change the situation. The first contender for the title of Earth 2.0 has always been Mars, cold and lifeless. At the moment, it is not yet possible to pack your bags and fly to Mars on a vacation for two weeks, but this is absolutely realistic and feasible, the question is only how soon. The most conspicuous minus of Mars (with the exception of the lack of a normal Internet) is the lack of an acceptable atmosphere for life. Fly to your destination, get out of the aircraft and breathe deeply, feeling all the freshness of the local air - this is not about Mars. But this will not always be.
Today we will meet with you a study that describes a new method for generating oxygen through an unusual chemical reaction. How did scientists produce molecular oxygen from CO 2 , how effective is this method, and where can it be used other than interplanetary travel? We will look for answers to these questions in the report of scientists. Go.
Study basis
A person needs only three things for life: food, water and air, in which there will be enough oxygen. These are our primary needs (yes, the Internet is not on this list). But oxygen just doesn’t lie on the road, especially outside of our beloved planet. Therefore, if we want to go somewhere, we need to take it with us and use various devices to clean the air and reuse it. All this joy takes a lot of places, and sooner or later oxygen will be exhausted. And replenishing reserves in space or on Mars will be, to put it mildly, problematic.
However, relatively recently, scientists discovered a fairly large amount of oxygen in comet 67P / CG6, which prompted them to ask a very obvious question - where did it come from? Oxygen in bodies such as comets is the result of an abiotic reaction that occurs under extreme conditions, during which from H 2 O, CO 2 , CO, etc. O 2 (oxygen) is released. Scientists claim that such reactions can explain the presence of oxygen in comets, the upper atmosphere of Mars and in the early atmosphere of the Earth. A person can use such a chemical reaction to produce O 2 from CO 2 , which will make Mars suitable for life.
If it is very exaggerated and brief, then the reaction mentioned above is the decomposition of CO2 on components: C + O 2 . In other words, dissociation.
The dissociation of CO 2 can take place in several scenarios depending on the energy available for the reaction. Partial dissociation of CO 2 → CO + O (5.43 or 7.56 eV) requires the least energy . There is also a complete dissociation of CO 2 → C + O + O, requiring 16.46 eV. And, the most curious, exotic dissociation is when CO 2 decomposes into C and O 2 . Calculations show that a similar reaction proceeds on the surface of the potential energy of the ground state, first forming a cyclic intermediate CO 2 compound [c-CO 2 ( 1 A 1)], which then transforms into a collinear intermediate COO ( 1 Σ + ) on the path to dissociation into C + O 2 .
Such a reaction is possible if the molecule is “bent” so that the two O atoms become as close as possible to each other. This requires 6 eV of internal energy. And, you must admit, no matter how strong men bend metal rods, it will be much more difficult to bend a molecule.
Transitions into electronically excited and anionic states of CO 2 can help bend the molecule . Scientists recall that recent experiments by their colleagues have shown that the use of VUV (vacuum ultraviolet) photoexcitation and electron attachment allows the dissociation of CO 2 to C ( 3 P) + O2 (X 3 Σ g -). However, no one has previously studied this exotic process at a sufficiently detailed level. And all because as a result of such experiments no ionized O 2 products were found . But, as we know, not finding something does not mean that it is not at all.
Therefore, in the study we are examining today, scientists applied ion beam scattering methods along with mathematical modeling to demonstrate a new way to activate direct reduction of CO 2 to O 2 with the detection of ionized O 2 products . This process contains a previously unknown intramolecular reaction pathway that occurs during active collisions of ions and CO surface2 . The most surprising is the lack of dependence of this reaction on the nature and temperature of the surface.
Research results
First of all, scientists demonstrate the formation of O 2 in hyperthermic CO 2 + / Au collisions by plotting the kinetic energy distribution of three scattered molecular ion products (CO 2 + , O 2 + and O 2 - ) for different incident CO 2 + energies (E 0 ) . At E 0 <80 eV, a very weak signal of scattered CO 2 + was detected ( 1a , graph on the left).

Image No. 1 The
peak energy of the outgoing CO 2 + is proportional to E 0, that is, there is a ballistic or pulsed rebound from the surface, which excludes physical spraying. Scientists believe that observing a “dynamic” CO 2 + signal is crucial because it is evidence that some CO 2 survives a surface collision. In addition, it is possible to determine the sequence of collisions of constituent atoms. In addition to CO 2 + , signals of scattered O 2 ions were also observed ( 1b , 1c / graph in the center and on the right). In this case, the energies of the released O 2 + and O 2 -They represent a large fraction of the energy of incident particles (57%) and uniformly increase parallel to E 0 in a wider range than the scattered CO 2 + . The maximum signal of O 2 ions was observed at E 0 ~ 100 eV.
Researchers call the discovery of O 2 ionic products surprising, since neither the atomization of surface O 2 nor the abstraction of the O atom can explain the formation of these ions. And all because both of these mechanisms would produce O 2 at much lower output energies than was observed. It is logical to assume that the very dissociation of CO 2 is involved here .
Both partial and complete dissociation of CO 2 is in good agreement with other detected ionic products (CO + , CO - , O + , O - and C + ). And the energy of the outgoing CO + , CO - and O - changes in parallel with the energy of the incident particles, which is consistent with dynamic formation in a surface collision.
But the peaks O + and C + show an extremely small dependence on E 0 , which indicates a different origin, i.e., spraying. Confirmation of complete dissociation are scattered products C +manifested at E 0 > 80 eV.
Next, scientists used kinematics to describe the scattering mechanism.
The binary theory of collisions (BPS) allows us to calculate the kinematic factor, defined as the fraction of the energy of the incident particles, held by the scattered product emerging from the surface. In the simplest model, CO 2 + is scattered as a whole molecule, i.e. as a solid sphere with an atomic mass of 44 Da. In this case, the BPS predicts a kinematic coefficient of 0.6349, which is rather weakly correlated with the data ( 2a ).
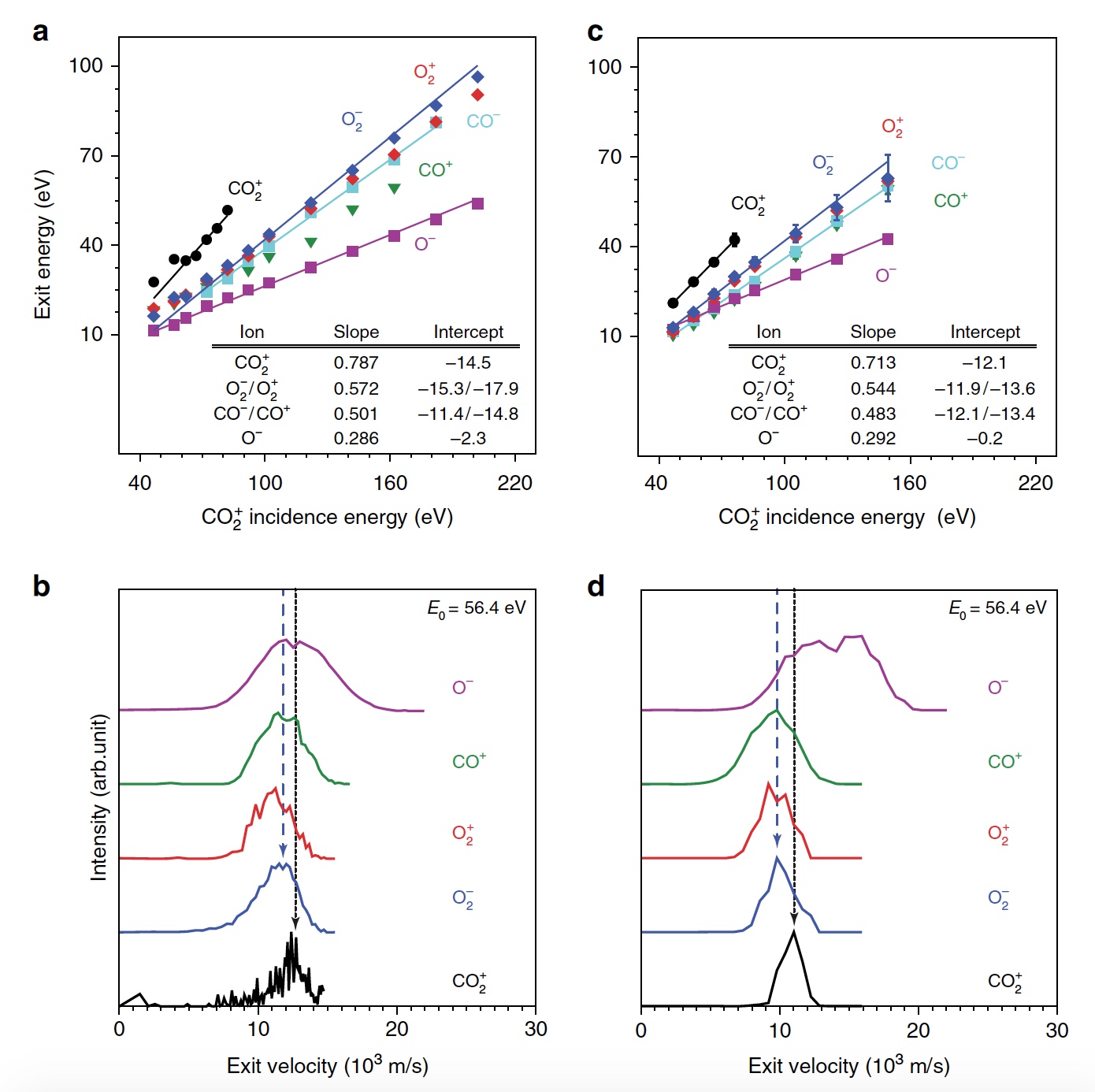
Image No. 2
After that, scientists applied a model in which the O atom first collides with the surface Au atom, and then the second collision of the CO fragment occurs without rapid dissociation of the CO 2 molecule . The application of BPS to this model of successive collisions gives a kinematic coefficient of 0.7870, which is in excellent agreement with the energy data of the outgoing CO 2 + (black line at 2a ). Graph 2a also shows the energies of other outgoing scattered ionic products.
The main potential source of such products can be called partial or complete dissociation of CO 2 and surface spraying of adsorbed CO 2 fragments. Despite the fact that some sputtering is indeed observed at high values of E 0 (> 140 eV), a kinematic analysis of the data on the energy of the outgoing particles provides convincing evidence of the presence of precisely pulsed dissociation of the CO 2 molecule .
Next, scientists conducted an analysis of the speed of the observed scattered particles.
Figure 2b shows a comparison of the peaks of the distribution of ions at E 0 = 56.4 eV. As we can see, the exit rates of scattered CO + , O 2 + , O 2 - and the slower part of the O - distribution overlap, which indicates a common origin. However, the distribution of O- noticeably wider, extending to higher output speeds, which may indicate an alternative origin. O 2 ion products exit at rates lower than CO 2 + due to inelasticity due to rupture of chemical bonds and non-resonant surface ionization.
The above kinematic analysis, scientists say, has provided conclusive evidence that some CO 2 molecules scatter intact after a two-step sequential collision of O and CO fragments. However, questions still remain regarding various aspects of the O 2 formation mechanism .
After the experiments, a number of questions remain. First, it remains unclear whether O 2 is produced by the electronic adiabatic or non-adiabatic mechanism. Secondly, the collision paths that underlie the velocity distribution at the exit of ionic fragments are unknown. Thirdly, it is not known how much neutral O 2 is formed .
In this case, modeling can clarify the situation, namely, the classical molecular dynamics method (MD method).
To simulate the scattering trajectory of CO 2 on Au (111), the scattering geometry observed in the experiment was applied. It is assumed that CO 2is released on the surface of the potential energy of a singlet electron in the case of neutralization of the incoming CO 2 + ions before a hard collision.
Quick neutralization occurs through resonant tunneling of electrons from the metal surface to the molecular cation, since the molecular level of CO 2 (-13.8 eV) is within the occupied Au zone (from -5.3 to -15.3 eV). In the simulation, electron transfers from / to the surface were also taken into account to take into account the ionization of neutral collision products.
Image 2c shows the calculated energy values of the outgoing particles.
It was found that a small amount of CO 2 maintains integrity at E 0> 80 eV, which is consistent with the absence of a signal at these energies during practical experiments. The data obtained through modeling are in excellent agreement with the experimental data, which is easy to see by comparing graphs 2a (experiment) and 2c (simulation).
In addition to this agreement between experiments and modeling, it also manifests itself in a comparison of the rate of output ions at E 0 = 56.4 eV ( 2d ). Both the model and the experiment show an expansion of the distribution of CO + and O - velocities . It was also confirmed that the distributions of O 2 + and O 2 -similar to the cation coming out slower than the anion. In addition, in both cases, it was found that CO 2 + emerges at a higher rate than ionized O 2 products .
Therefore, the use of this modeling technique can provide fairly accurate data regarding the reaction mechanism of the direct conversion of CO 2 to O 2 .
During the main stage of the simulation, 20,000 CO 2 -on-Au collision trajectories were recreated at different energies of the outgoing particles. As a result, several variants of dissociation products were obtained, including O 2 ( 3a ).
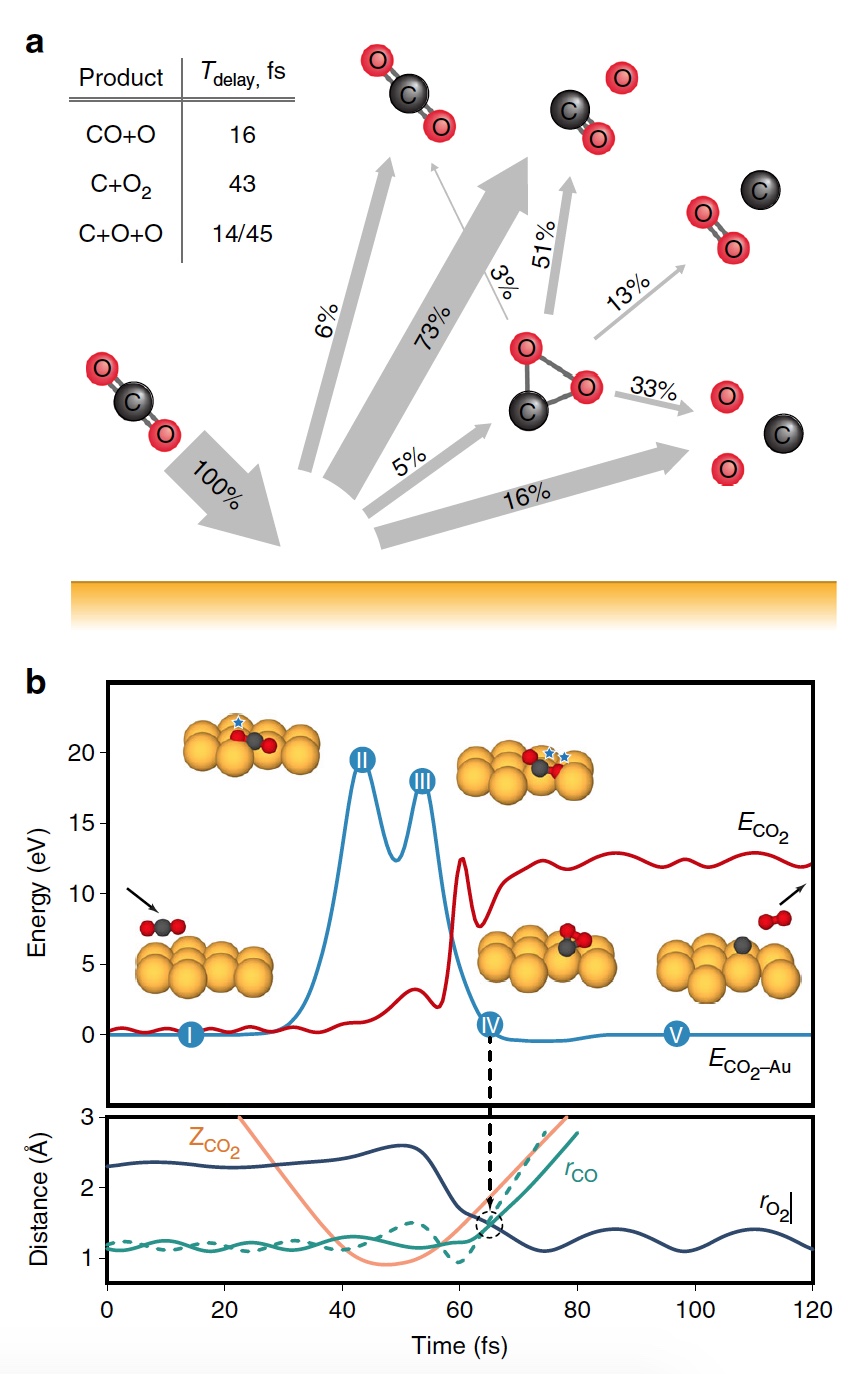
Image No. 3
On the image3b shows a representative trajectory resulting in the formation of O 2 . As a result of pulsed energy transfer during a collision , a significant intramolecular rearrangement occurs inside bouncing CO 2 . The O – O distance decreases, and the C – O distance increases, reaching a peak when CO 2 acquires a triangular configuration with almost equal bond lengths. Such a strongly bent intermediate CO 2 product has a significant amount of internal energy and quickly dissociates, resulting in the formation of a free C atom and a vibrationally hot O 2 molecule .
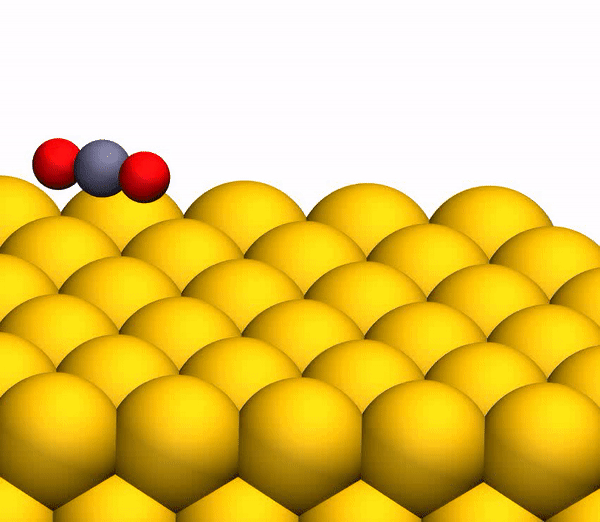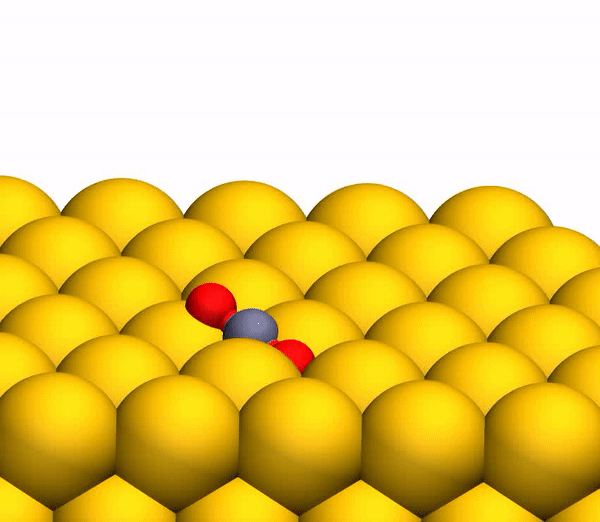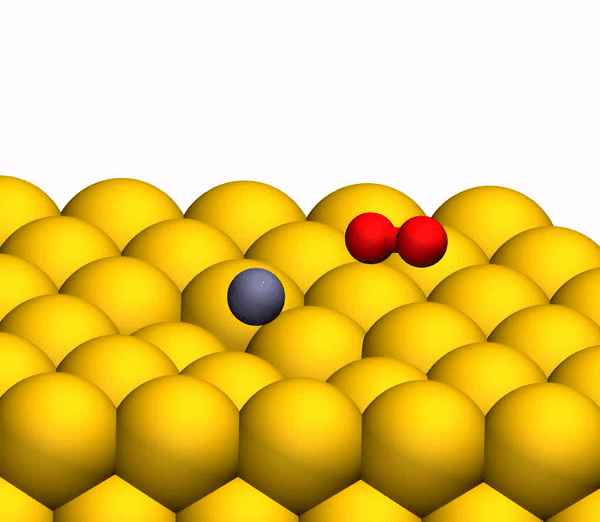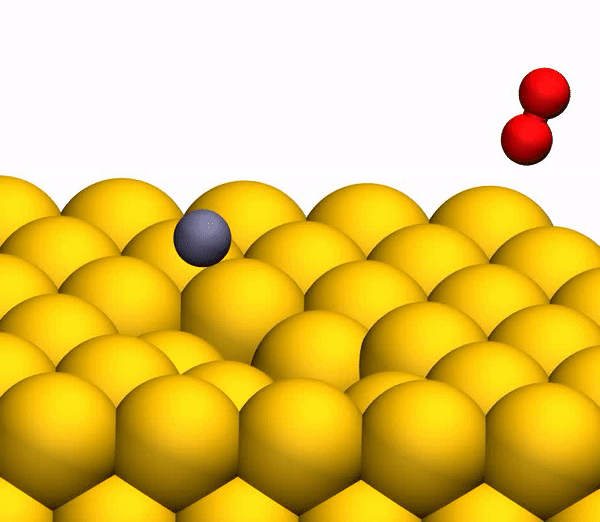
CO 2 separation processto a free C atom and a vibrationally hot O 2 molecule .
It is worth noting that the separation mechanism described above is fully consistent with the conclusions of the kinematic model used previously.
Calculations showed that approximately 5% of all trajectories lead to a strongly bent intermediate state of CO 2 , which is one of the stages of O 2 extraction . This condition is fragmented mainly through partial dissociation (51%), after which complete dissociation follows again, but with a higher result (33%).
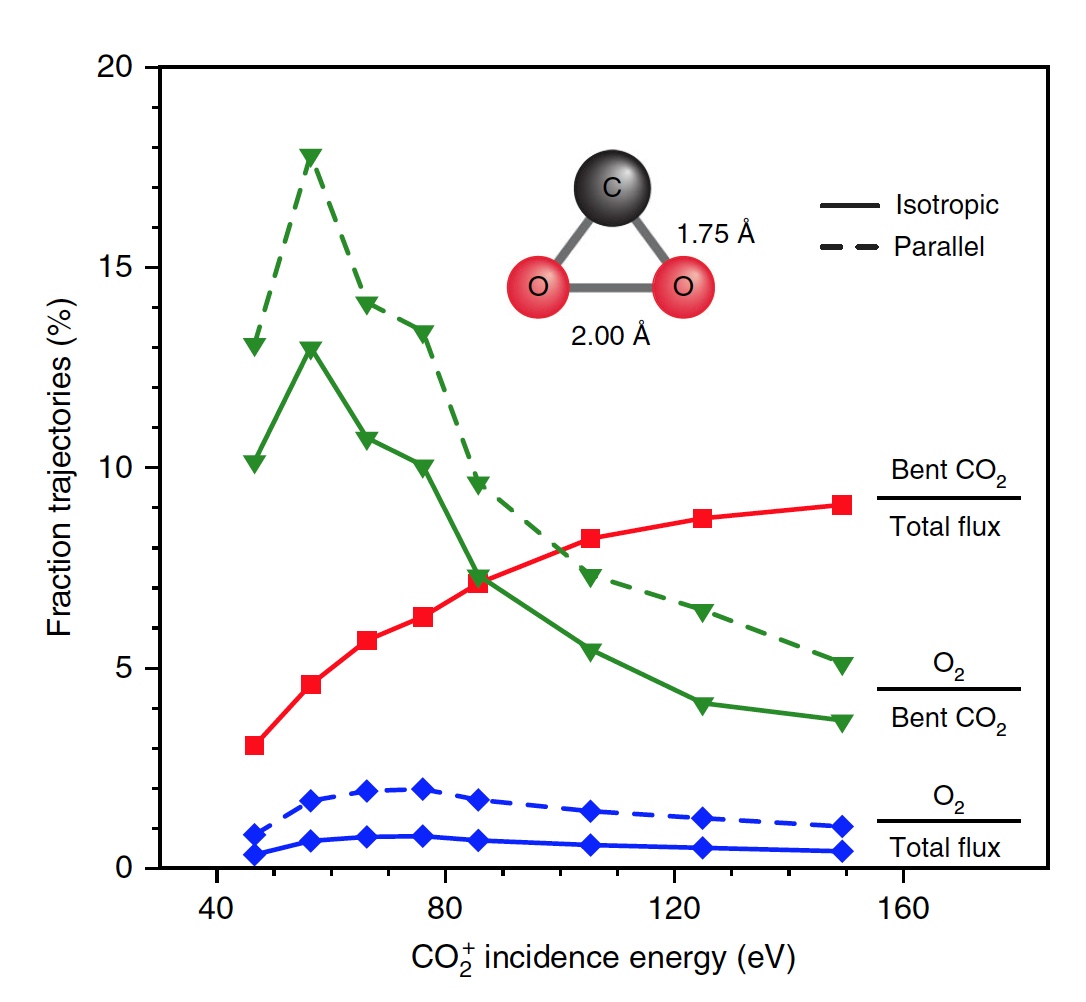
Image No. 4
Scientists note that of all curved CO 2 molecules, only 13 percent produce oxygen. That is, the overall result of CO 2 dissociation→ C + O 2 is 0.6% at E 0 = 56.4 eV. If you increase the energy of the incident particles, then you can get O 2 up to 0.8 ± 0.2% at E 0 ~ 70 ± 15 eV (blue line in image No. 4).
In addition, the fraction of O 2 -forming trajectories increases significantly when a strongly bent intermediate state of CO 2 (green line) is reached, reaching a maximum of 13% at E 0 ~ 55 ± 10 eV. The lower total yield of neutral O 2 is due to the fact that only a small fraction of linear CO 2 molecules reaches a strongly bent state (red line). If you specifically change the orientation of the incoming CO 2 molecules(axis parallel to the surface), then as a result, you can get 2% more O 2 (dashed green line).
For a more detailed acquaintance with the nuances of the study, I recommend that you look into the report of scientists .
Epilogue
This work has demonstrated that through non-standard chemical reactions it is possible to obtain oxygen from carbon dioxide. No one will argue that such a technique requires further refinements and improvements. However, the fact that this work really opens up new possibilities for interplanetary travel, the colonization of Mars, and not only cannot be denied. There is also a problem with oxygen and carbon dioxide on our home planet. Rather, with the latter, for its volume has increased significantly since industrialization. And given the fact that CO 2is one of the main culprits of global warming, reducing its amount and getting oxygen from it sounds like a very tempting idea. Since we still can’t do without factories, factories, cars with internal combustion engines and other things, the new methods of cleaning our atmosphere are not just needed, but extremely necessary. For travel to distant planets is certainly cool, but before the massive move to Mars is still very, very far, therefore it is worth thinking more about the Earth on which we live than about Mars, about which we dream.
Thank you for your attention, remain curious and have a good working week, guys! :)
Thank you for staying with us. Do you like our articles? Want to see more interesting materials? Support us by placing an order or recommending to your friends,30% discount for Habr users on a unique analogue of entry-level servers that was invented by us for you: The whole truth about VPS (KVM) E5-2650 v4 (6 Cores) 10GB DDR4 240GB SSD 1Gbps from $ 20 or how to divide the server correctly? (options are available with RAID1 and RAID10, up to 24 cores and up to 40GB DDR4).
Dell R730xd 2 times cheaper? Only we have 2 x Intel TetraDeca-Core Xeon 2x E5-2697v3 2.6GHz 14C 64GB DDR4 4x960GB SSD 1Gbps 100 TV from $ 199 in the Netherlands! Dell R420 - 2x E5-2430 2.2Ghz 6C 128GB DDR3 2x960GB SSD 1Gbps 100TB - from $ 99! Read about How to Build Infrastructure Bldg. class using Dell R730xd E5-2650 v4 servers costing 9,000 euros for a penny?
