DIY Oxy-Acetylene Welding
 The path to IT is very thorny for everyone. For example, in childhood, I wanted to be a welder - it's so beautiful when splashes of molten metal fly around! But somehow it didn’t work out: they started to write out the Young Technician magazine, where on the last page of one of the numbers they talked about the robot controlled by the BK-0010 computer ... But the fad remained ...
The path to IT is very thorny for everyone. For example, in childhood, I wanted to be a welder - it's so beautiful when splashes of molten metal fly around! But somehow it didn’t work out: they started to write out the Young Technician magazine, where on the last page of one of the numbers they talked about the robot controlled by the BK-0010 computer ... But the fad remained ... Also, someone probably remembers the program "Crazy hands" where various creative (as we would say now) things were made from plastic bottles.
Under the cut, I’ll show you how to make a real oxygen-acetylene welding from a plastic bottle, an insulin syringe, several meters of rubber hose, a glue gun (where without it) and some other things that can be found in every home *.
* In every BarsMonster house .
Theory
 The flame temperature depends on the heat of combustion of the fuel and the heat capacity of the reaction products. When we burn something in the air, we also have to heat nitrogen (which is almost 80%), because the temperature of the flame in the air is usually not high (~ 1500-2000C and lower). But in pure oxygen, with the correct ratio of fuel and oxygen volume, only reaction products need to be heated, and much higher temperatures are achievable.
The flame temperature depends on the heat of combustion of the fuel and the heat capacity of the reaction products. When we burn something in the air, we also have to heat nitrogen (which is almost 80%), because the temperature of the flame in the air is usually not high (~ 1500-2000C and lower). But in pure oxygen, with the correct ratio of fuel and oxygen volume, only reaction products need to be heated, and much higher temperatures are achievable. As a fuel, hydrocarbons are usually considered. Carbon gives carbon dioxide during combustion, and hydrogen gives water. Water has a very high heat capacity (4.183 versus 1.4 kJ / (kg * K)), respectively, the more carbon there is in the fuel and less hydrogen, the higher the potentially achievable temperature as a first approximation.
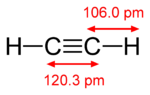
The best combination is in acetylene C 2 H 2, for example, methane CH 4 and propane C 3 H 8 - this ratio is much worse.
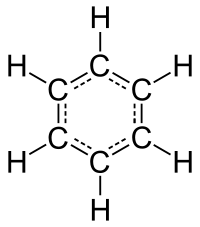 But there are other compounds with an equal amount of carbon and hydrogen - for example benzene, C 6 H 6 . In addition to benzene toxicity, less energy is released during its combustion, as in acetylene, the "excess" energy is stored in an unstable triple carbon bond, which provides it with one of the highest combustion temperatures in oxygen - 3150 ° C.
But there are other compounds with an equal amount of carbon and hydrogen - for example benzene, C 6 H 6 . In addition to benzene toxicity, less energy is released during its combustion, as in acetylene, the "excess" energy is stored in an unstable triple carbon bond, which provides it with one of the highest combustion temperatures in oxygen - 3150 ° C.This excess energy (~ 16%) can be released during spontaneous detonation of compressed acetylene even without air access (the reaction product is just benzene and vinylacetylene). Wikipedia claims that this requires a pressure of only 2 atmospheres - but I squeezed acetylene into a syringe to 4-5 atmospheres and nothing happened (apparently I need catalysts, shock or elevated temperature). In any case, because of this effect, acetylene is not stored in compressed form, but is dissolved in cylinders in acetone. But there is also a simpler and safer method for producing acetylene with small volumes - the reaction of calcium carbide with water. This method will be used.
What is noteworthy, you can achieve even higher temperatures - if you use substances that do not contain hydrogen at all as a fuel: cyanogen(hello Android), (CN) 2 - burns at 4525 ° C and dicyanoacetylene C 4 N 2 , burns at 4990 ° C (again thanks to triple carbon bonds, and a smaller relative amount of excess nitrogen). But practically for this purpose they are not used due to toxicity.
Security
Compressed oxygen and acetylene in cylinders can be very dangerous with the slightest violation of the operating rules, because of course I will not use them.Acetylene will be generated from a small amount of calcium carbide (~ 100g per session), in a 0.5l bottle. Initially, I wanted to use 2L so that the pressure was more even - but after watching on YouTube how a liter of acetylene with oxygen explodes , I decided to cut sturgeon. To avoid creating a dangerous pressure in the generator, the output of acetylene on the burner should never be blocked. The acetylene generator needs to be cooled - otherwise there will be "self-acceleration" of the reaction due to heating.
Oxygen - will be generated by a medical oxygen concentrator, which is relatively safe.
There could still be a danger of pumping oxygen into the acetylene generator, followed by popping - but for this it is necessary that the protective valve in the oxygen generator does not work and the gas outlet from the burner is blocked (for example, by dirt).
And of course you need to work with special glasses - not only to protect against metal splashes, but also ultraviolet radiation from the flame (i.e. transparent plastic safety glasses will not work here).
To prevent the accumulation of explosive concentrations of acetylene in case of leaks - the fan constantly blew around the workplace + all operations were carried out in the open air.
There is also the problem of a “reverse blow”: when the gas flow rate in the burner becomes too low, the flame goes inside the burner with cotton, and if there is air in the acetylene, the flame can reach the acetylene generator. Therefore, I did not set fire to acetylene immediately after the start of the reaction, but I waited ~ 15-30 seconds until the air was expelled. Also, this problem can be solved by adding a water valve to the acetylene pathway.
Design
So, we need an oxygen generator. In my case, the Atmung medical oxygen concentrator (the price is about 20k rubles - but, fortunately, it was already in stock). It can generate 1 liter per minute 95% oxygen, and larger volumes with a decrease in concentration. It works on the principle of short-cycle non-heating adsorption - due to the different speeds of the passage of gases through the pores of the zeolite:
Next - the standard acetylene burner "Baby", it has the smallest nozzle, purchased in an online store (960 rubles):

My acetylene generator works as follows: water from a can standing at a height of 1-2 meters (to create pressure) through a needle of an insulin syringe drips small drops onto calcium carbide in a bottle. As soon as the pressure rises due to the released gas, the water stops dripping, until the pressure drops. Thus, the system stabilizes itself. Nevertheless, the generator in a jar of cold water - to prevent excessive heating:

Result
The flame of acetylene in the air smokes a lot, and it looks quite ordinary:
With the inclusion of oxygen, everything changes:

You can melt and set fire to steel, but still there is not enough power (you need to take a thicker tip, increase the pressure):

It turned out that a flexible glass fiber optic turns out automatically - when the molten glass drips, as soon as the neck thickness becomes sufficiently small, it cools very quickly and does not thin out further.

You can melt glass like oil, seal capsules from glass tubes:

The task of life is completed, I hope you were interested :-)
PS. And do not repeat it at home.
Addition from a specialist (@freuser):
From the point of view of a professional welder (30 years, 11 years of experience, 2 of them are gas welding):
Good article, in general the disclaimers are correct. It is worth adding that work is being carried out on fireproof surfaces (sparks fly 2 meters from the wind, and metal droplets even darkened to ordinary colors may burn shoes if they are shoes.)
The design of the generator is called VK (water on carbide), there is also KV and VV (google with schemes, copyright is still Soviet :)).
There are no comments on the video, there’s nothing special to watch (from my point of view), only it’s worth adding that large glass (or whole bottles), as well as stone / concrete / some bricks can burst / delaminate when heated, forming low-flying fragments, which are wonderful they penetrate and fuse into the skin (especially on the face), however, by a millimeter, no more, and are easily removed from there.
I would also like to answer it on habrahabr.ru/post/185720/#comment_6461342: this is not a return blow, or rather, not what Nepherhotep warned against, but simply the burner either overheated, or rather, from low pressure and an obstacle close to the nozzle (or a blockage inside the nozzle) the flame went towards the flow, to the injector (in this burner it is under the union nut, between it and the valves), but it did not move further. And usually, by a reverse blow we mean the case when the flame slipped through the injector and went through the hose towards the source. There are two types of backward strokes (one I saw with my own eyes): the flame goes along the acetylene hose (normal combustion, only the end of the hose constantly burns and the flame moves uniformly to the cylinder / generator) and oxygen (everything is prettier here - the hose is suddenly 20-30 cm flashes with a piece and turns into rags, a second pause - the next segment, etc. until the tank itself.) Although the second case is rare. The simplest protection is to squeeze the hose in the distance, press down with your foot (do not forget about the shoes) and yell to your partner “Sanka, close the cylinders, *** !!” For more civil protection, you can make water locks - also a bottle, two tubes, one to the bottom - incoming, the second short - to the burner. Up to half is poured with water and that's it, the bubbles run beautifully))
