Radiation: the invisible killer and his daughters or a little about radon

In previous articles and their discussion, I have repeatedly argued: no modern methods can reliably detect the effect of the magnitude of the natural radiation background in a wide enough range of it on human health. But there is one natural radiation factor, the influence of which is relatively clearly visible. This is a radioactive inert gas radon , nicknamed by hunters to the red word by journalists as the "invisible killer."
Radium emanation
In 1899, Rutherford and Owens discovered that in addition to radioactive radiation, thorium emits a certain substance, which also possesses the main property of radioactive radiation - the ability to ionize - behaves like a gas: it is transported with an air current, and does not propagate in a straight line, diffuses through porous media, delayed by the thinnest solid partitions, and in addition, it “settles” on objects placed in its environment, telling them radioactivity rapidly falling off by the exponential law. This was unusual: before that, radioactivity seemed to be an extremely constant phenomenon. Simultaneously with them and not knowing anything about their work, a similar phenomenon was observed by the German Friedrich Dorn, who worked with radium and also emitted radioactive gas from it. The gas released from radioactive substances was called emanation.
Rutherford and Soddy, who joined him, began to clarify the nature of emanations. In the spectrum of a gas discharge in emanation, helium lines were present. Moreover, their intensity rapidly increased simultaneously with a decrease in the radiation intensity from the tube with emanation. The relationship of helium with radioactive minerals was already known: on the ground it was first isolated from minerals containing thorium. When in 1903 it was possible to collect a sufficient amount of emanation, it was possible to see the spectrum of emanation itself, which was different from the spectra of all other gases. It was not a spectrum of helium: it was a spectrum of a new chemical element.
The emanation was not helium. But she turned into him! Its spectrum weakened over time, and in its place appeared the familiar spectrum of helium with its yellow line next to the sodium doublet. It was something new and incredible: scientists watched how, before their eyes, one chemical element turned into another.
The most difficult task fell on the role of U. Ramzai: he managed to isolate a tiny amount of new gas in free form and he managed to determine its density. The molecular weight calculated from it turned out to be 222, which was less than the atomic mass of radium by exactly four — the atomic mass of helium.
It turned out that radium turned into helium and emanation. And then emanation turned into helium - and something else.
Further research of Rutherford identified alpha particles with helium atoms, and the picture finally developed. The fact of the existence of a fundamentally new natural phenomenon - the transformation of some elements into others with the emission of rapidly flying particles - was reliably established. And it broke all the scientific ideas that barely had time to take shape. Not so long ago, the concept of an atom was formed - an elementary indivisible and unchanging unit of matter, as it turned out that an atom can suddenly decay, and its “fragments” will be two new atoms of other chemical elements.
And emanation, meanwhile, through the efforts of Ramsay, took its place in the periodic system, adding another element to the inert gas family and was later renamed radon.
Radon as a substance
From a chemical point of view, radon is an inert gas. Like xenon, it is not as inert as helium, neon or argon, and unlike the latter, it has some chemical properties . However, in ordinary life they can be safely neglected: the ability of radon to enter into chemical compounds is too small. But it is easily adsorbed by tissues, paper, activated carbon and silica gel, it dissolves in oils and from a solution in water actively passes into ice when freezing, forming clathrates. Radon also forms stable clathrates with a number of other molecular compounds - for example, radon clathrate with glucose is well known and used in "radon medicine".
Pure radon glows due to radioactivity. Especially bright - blue light - glows liquid radon, which freezes upon further cooling and when approaching the temperature of liquid nitrogen changes the color of the glow to yellow, and then to orange. As the decay products accumulate, liquid and solid radon, which is initially colorless, darkens.
But outside special laboratories and hot chambers, we will never see either liquid or solid radon. Even gaseous, it is found in nature only in a very small concentration. After all, a gram of radium per day forms everything
Radon as a radionuclide
In total, 19 isotopes of radon are known, but only two isotopes of radon can be encountered in ordinary life: actually radon (emanation of radium) with an atomic mass of 222 and a short-lived thoron with a half-life of 55 seconds and a mass number of 220. There is also a third natural radon isotope of actinonIs a short-lived member of a series of uranium-235-actinium, but due to the short half-life and low content of uranium-235 and its "daughters" in nature it is difficult to detect. Radon-222, having emitted an alpha particle with an energy of 5.59 MeV, turns into polonium-218 (often denoted by the old Curie spouses, the designation RaA) with a half-life of only 3.1 minutes, and he again “spitting out” alpha -particle turns into lead-214 (RaB), or undergoes beta decay, turning into astat-218 and almost immediately - through alpha decay - bismuth-214 (RaC). Lead-214 also turns into the latter. Lead and bismuth-214 have half-lives of slightly less than half an hour and their atoms, formed after the decay, have time to condense during this time, forming the so-called active plaquecovering the surface of dust particles and other aerosol particles. Beta activity makes such dust particles positively charged. Bismuth-214, having emitted a beta and alpha particle almost simultaneously (via polonium-214), passes into a rather long-lived (22 years) lead-210, on which a fast chain of transformations is suspended. The alpha decays of polonium-218 and polonium-214 account for the majority of the internal dose caused by radon-222. But the dose from radon itself does not exceed 2% of the total dose.
This chain of radionuclides that quickly transfer into each other - polonium-218, lead-214, bismuth-214, polonium-214, lead-210 - is called the daughter products of decay (DPR) of radon and is inextricably associated with it in the air. Together with radon, we breathe them into our lungs, and when it rains, it flushes them out of the air, because of which rainwater acquires radioactivity with a half-life of approximately 25 minutes. This radioactivity can be easily detected by wiping any surface with a rag in the rain and measuring the rag with a household dosimeter, preferably with a mica sensor (the lead cover on the sensor must be removed). At the same time, many take the shocking testimony of the dosimeter for the consequences of the Chernobyl disaster, Fukushima or the signs of some kind of accident that the authorities are hidingbut in fact the reason for this is radon. An increase in the radiation background during heavy rains is partly associated with it (and partly with the scattering of cosmic muons on raindrops with the formation of secondary electrons and bremsstrahlung gamma radiation).
Thoron, on the other hand, lives less than a minute and usually breaks up almost where it formed. Having emitted two alpha particles in a row (through the living fraction of a second polonium-216 - thorium-A), it turns into lead-212 (thorium-B), which lives for 10 hours and forms an active plaque of thoron together with its “heir” bismuth-212 ( thorium-C) with a half-life of 1 hour. The latter makes a “fork”: in one of its branches, having emitted an alpha particle, it turns into thallium-208, famous for its 2.6 MeV gamma line on the far right on the energy scale, and through beta decay, it turns into polonium-212, which instantly (in microseconds) emits an alpha particle of very high energy (10.5 MeV). In both cases, stable lead-208 is formed. Due to the short life time, the toron practically does not have time to fly apart and we do not breathe it.
As a characteristic of the radon content in air, a value called equivalent equilibrium volumetric activity (EERA) is usually used. It is calculated for radon-222 by the formula:
Where
Similarly, according to the formula
determine the ERAA of radon-220. Here ThB and ThC are, respectively, lead and bismuth-212.
Here
In the future, speaking of "concentration", "level", "content", etc., I mean exactly EROA.
Killer radon (and a little healer)
The decay of radon-222 and its daughter products is due to approximately half the dose of natural human exposure. As practically the only natural radionuclide present in the environment in the form of gas (not counting the negligible amounts of tritium and radiocarbon), radon almost completely forms the dose of radiation to the lungs from the inside. The lungs are an organ of relatively high radiosensitivity due to the constantly renewed epithelium of the alveoli, so the risk of lung cancer when they are irradiated is about three times higher than the overall risk of oncology with uniform exposure to the body. And after the decay of radon, its DPR (and hereinafter - polonium-210, formed from the lead-210 remaining in the lungs, which has the ability to accumulate in the lungs) is fixed in the lung tissue and irradiated with alpha particles, each of which having an energy of 5- 6, and for a thoron - up to 10 MeV, and a quality factor of 20, is a very destructive "shell". For each atom of radon of such "shells" there are four pieces, and for the atom of thoron - three.
Because of this (and also because lung cancer in non-smokers is a rare occurrence), even relatively low levels of radon concentration are reflected in the incidence of lung cancer. According to the US Public Health Service, radon is the second cause of tumor incidence of this localization after smoking. At a concentration of radon in air of 200
It is also believed that radon, in addition to the well-known stochastic effects, also provokes cardiovascular diseases. However, this opinion is usually expressed in connection with an attempt to explain the so-called geopathogenic zones, the existence of which is in itself quite doubtful.
In general, it is radon that is currently the most important problem of protecting the population from a radioactive threat. This is especially true in some regions where radon is actively emitted from the bowels of the Earth and its concentration in the basements and on the first floors of buildings is extremely high.
Such a place on Earth, for example, are the Caucasian Mineral Waters, Beshtau. To assess how serious it is, I recommend watching this video:
Can you imagine what will happen to the lungs of someone who snoops there without respiratory protection?
The same situation as in the Caucasian Mineral Waters is observed in other regions known for their granite massifs, volcanoes, hot springs and uranium ores - Switzerland, Austria, the Czech Republic, on a smaller scale - Finland and northwestern Russia, as well as southern Siberia, Far East. In these regions, urgent need is measures to reduce the concentration of radon in residential premises - radon protection.
The map below shows the doses received from radon by residents of various regions of Russia (in mSv / year).
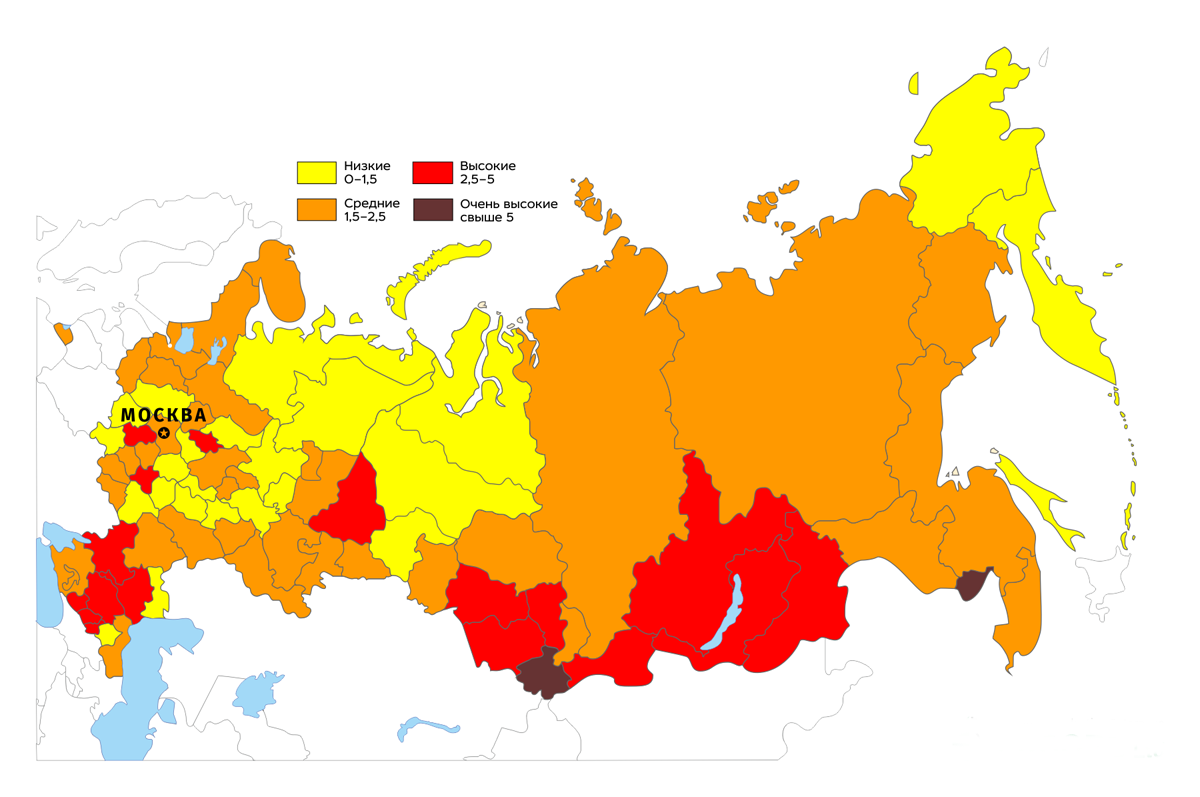
However, there is an opinion that the radon problem is exaggerated. The above figures for cancer incidence are not experimentally established, but calculated, based on data on the incidence of people living and working at significant levels of radon - miners, workers and residents of radon resorts, etc. At the same time, the non-threshold concept, on the basis of which these figures are calculated, has not been experimentally proven and remains a hypothesis, albeit well-founded theoretically. As an argument, the well-known therapeutic effect of radon in various diseases is usually indicated. It is known that radon has an analgesic and anti-inflammatory effect, it causes (probably, through increased production of DOPA and related biologically active compounds by skin melanocytes) activation of a number of neuroendocrine mechanisms, giving a pronounced effect on the cardiovascular and nervous systems, and also enhances microcirculation in the irradiated skin. Radon baths have been shown to be effective in many diseases.
In addition, there is evidence that the alpha radiation of particles coated with "active plaque" stimulates the activity of pulmonary cilia, helping to remove these particles from the lungs, and this mechanism can significantly reduce the effect of low concentrations of radon.
Despite the fact that thorium (in activity) is no less than that of uranium, the proportion of thoron in the total dose is only about 5%. This is due to the fact that he "does not live out" to our lungs, in most cases, simply not having time to reach the surface.
Sources of Radon
The half-life of radon-222 is only 3.8 days, but due to its constant formation during the decay of radium, new radon is constantly entering the atmosphere. Sources of radon, therefore, are uranium-rich rocks, mainly granites, but much more active and uranium-rich rocks are also found. So, phosphorites are known for their uranium bearing. But the greatest amount of radon is emitted not from the monolithic granite massif, but from the faults leading to the bowels of the Earth, forming the so-called "radon breath". The allocation of radon is a kind of marker by which such faults can be found, and hence deposits of various minerals confined to them. Radon is especially intense in volcanic regions. Sometimes they find an intense release of radon in places where, it would seem, from nowhere. A detailed study reveals a deep fault. And the intensity of radon emission is rich and, most importantly, a fairly quick source of information about changes in the state of the earth's interior. Its vibrations portend earthquakes and volcanic eruptions, allow predicting mountain impacts in mines, and help prevent accidents when drilling wells.
Radon is also released from building materials. The “leader” here is phosphogypsum - a material obtained as waste from the production of phosphate fertilizers, which concentrates a significant part of the radium contained in the original phosphorite (in which there is a lot of it, like uranium), so that radon phosphogypsum releases a lot. And since the disposal of phosphogypsum is a real problem, the temptation to use it as gypsum in the composition of building mixtures is very high. So there are "plumbing" and radon-emitting gypsum boards, self-leveling floors and plaster.
I already talked about the radioactivity and “radonogenicity” of granite - while granite gravel and sand often become a component of concrete used in construction. In this case, it is necessary to be guided by NRB-99 and use varieties of granite with different radioactivity, where this is permissible. Granite is usually divided into 4 classes of radioactivity:
I - up to 370 Bq / kg - allowed to be used without restrictions in any construction,
II - up to 740 Bq / kg - can be used in non-residential buildings (including public) and for exterior cladding,
III - up to 2800 Bq / kg - only for road construction outside settlements,
IV - up to 3700 Bq / kg - can be used in construction only where it will be covered with a thick layer of low-activity material.
With an activity of more than 3700 Bq / kg, granite is not used in construction.
Moreover, for the preparation of concrete for residential buildings, only the most low-grade granite of radioactivity class I is used.
Ceramic tiles and granite cladding may also be a source of radon in rooms. But usually these sources can be neglected. By the way, uranium glass, which some Russian celebrities (and not only they) like to collect, is not a source of radon danger: radon is not only unable to go beyond the solid mass of the glass, but practically does not form in this glass, since it very little radium. When uranium was extracted from the ore, the radium contained in it was removed, and the new one did not have time to form. But samples of uranium minerals and devices with continuous light composition based on radium-226 can “radiate” an apartment to quite dangerous levels.
In radon-hazardous regions, the strongest source of radon is the water supply, if the water for it is taken from artesian wells. So, while taking a shower, the concentration of radon in the room can rise from 50-100 Bq / m ^ 3 to several kilobecquerels per cubic meter. Gas also supplies radon to our apartments.
Radon danger is sharply aggravated ... energy saving. It forces you to make houses much more airtight than before, to ventilate less and less, to actively use air recirculation, which means that the radon that has got into the room remains in it. Therefore, materials and construction approaches that in our country lead to acceptable levels of radon, as the fight against heat leaks intensifies, can give serious growth.
Detection and measurement
How do you know what the level of radon is where you live or work? Unfortunately, this is not very simple. Although radon is the source of half of the natural background radiation, the “normal” readings of the dosimeter are not at all a sign of well-being. In general, radon can be detected by a dosimeter in rare cases of very high levels - while its characteristic feature is smooth, wave-like fluctuations in dose rate and a rapid decrease in radiation levels when opening doors and windows.
There are a number of “standard” methods used for official measurements that quantify radon content. The first of these is the direct calculation of alpha decays in an ionization chamber filled with the test air. Decays are recorded by very weak current pulses that occur when charges formed during the passage of an alpha particle, or by the ionization current, which is usually not measured directly because of its extremely small value, but determines the discharge time of the structural capacity of the ionization chamber. Another method is scintillation - a zinc sulfide layer deposited on the hemispherical inner surface of the working volume is used as a scintillator, and the PMT is the “plug” covering the detector. Semiconductor alpha radiation sensors are used similarly, but because of the short path, it is impossible to make a detector for a large volume of gas, and the time for measuring ordinary radon activities (tens of Bq / m ^ 3) is stretched for many hours, or even a day. Significantly reduce the measurement time by collecting radar DPR on the surface of the detector electrostatically: such well-known devices as SIRAD MR106N, Radex MR107 work this way. These are inexpensive devices, the cost of which is comparable to the price of simple dosimeters (about 10,000 rubles). Unfortunately, in such devices, long-lived decay products (lead and polonium-210) accumulate over time on the detector, gradually increasing the background hardware, especially when using such devices in radon-infected rooms, which requires replacement. Significantly reduce the measurement time by collecting radar DPR on the surface of the detector electrostatically: such well-known devices as SIRAD MR106N, Radex MR107 work this way. These are inexpensive devices, the cost of which is comparable to the price of simple dosimeters (about 10,000 rubles). Unfortunately, in such devices, long-lived decay products (lead and polonium-210) accumulate over time on the detector, gradually increasing the background hardware, especially when using such devices in radon-infected rooms, which requires replacement. Significantly reduce the measurement time by collecting radar DPR on the surface of the detector electrostatically: such well-known devices as SIRAD MR106N, Radex MR107 work this way. These are inexpensive devices, the cost of which is comparable to the price of simple dosimeters (about 10,000 rubles). Unfortunately, in such devices, long-lived decay products (lead and polonium-210) accumulate over time on the detector, gradually increasing the background hardware, especially when using such devices in radon-infected rooms, which requires replacement.
A filtration method is also used. Several cubic meters of air are pumped through the sorbent layer and then the radioactivity of the sorbent is measured. To do this, use a gamma spectrometer, recording peaks of lead and bismuth-214. There are specialized devices that include a detector with a gamma spectrometer and a pump with a filter cell, housed in one housing. These are expensive instruments that allow for a short time to determine the minimum activity of radon and track small fluctuations in the ERAA of radon.
The simplest version of this method is not difficult to detect the presence of radon in the apartment - for this it is enough to use a vacuum cleaner and a Petryanov filter (any respirator), and then measure the filter using a dosimeter with a mica sensor. But to quantify it, you need to standardize the technique and carry out the calibration. And this is already practically unavailable at home. But if, after several minutes of operation of the vacuum cleaner, the dosimeter showed a much larger value than the natural background, this is a reason to sound the alarm.
The same applies to the well-known “radon trap” method. The trap itself is simple to manufacture: it consists of a voltage multiplier with an output voltage of minus 600-1500 V and a metal plate or grid to which this potential is supplied. The scheme of the multiplier given by the notorious Oleg Aizon looks like this:
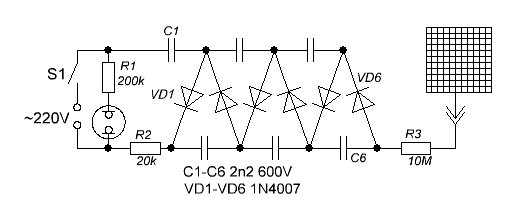
(the scheme is taken from the forum, in the same place - almost everything about its manufacture and use). An electrode under negative potential is placed in the measured room and left there for 6-8 hours, and then it is measured by a radiometer with the gamma filter cover open.
The mechanism of operation of the radon trap is due to the fact that aerosol particles coated with active plaque of radar DPR acquire a positive charge due to beta activity and are attracted to a negatively charged electrode. After some time, between the precipitation of new DPR radon and the decay of the already settled, equilibrium is established at which the activity of the deposited DPR is proportional to the concentration of radon.
Oleg Aizon gives the following "reference points of the scale":
10-60 μR / h - normal radon level,Of course, these figures will significantly depend on what the measurement is made: the Arizona-Stora-TU radiometer used by the SBM-20 meters will give lower readings than a radiometer with a mica sensor, for example, the MKS-03CA.
70-150 μR / h - increased radon level of
150 μR / h or more - there is a radon source in the room
400-600 μR / h - very high radon content
Of the other "professional" methods for determining radon, track detectors should be noted. The detector itself is very cheap - it is a polycarbonate film coated with a layer of filter material that does not pass radon and other radioactive dust to the DPR film, but does not trap radon itself. The film is left for a certain time in the test room, shaft or well, and then "appears" by etching. The areas destroyed by alpha particles dissolve in the etchant and pits remain on the film, the amount of which is proportional to the concentration of radon multiplied by the exposure time. In some countries, such detectors are distributed among residents of radon-hazardous regions with instructions and instructions to send to a specific address after exposure.
* * *
Contrary to popular belief that “everything natural cannot be harmful,” radon is possibly the cause of more deaths than smoking, car accidents, and household accidents. So protection from it in radon-hazardous regions is urgently needed. Whether radon is harmful at relatively low levels is an open question.
