Bubble Physics: A Search for Foam Destruction Mechanism

The simplest things can have the most unusual and even unexplored aspects. From an early age we try to understand the nature of everything that surrounds us. How the light in the chandelier works, why the sky is blue, where it rains, why the lemon is sour, and sugar sweet - this is just a small list of questions that a curious child can ask in a very short period of time. Growing up, we are not so much interested in such things, paying attention to something more important, in our opinion. But understanding the nature of simple, at first glance, things can be of great benefit.
Today we will get acquainted with a very unusual study in which scientists tried to understand the mechanism of foam destruction. Have you ever wondered why the foam in your cappuccino is not as durable as we would like? If you were told that you simply do not know how to cook it, now you will have a very scientific counterargument. Which sequence of events leads to the destruction of the foam structure, what is the catalyst for this process and what is the use of such knowledge? We will find answers to these and other questions in the report of the research group. Go.
Study basis
No matter how simple the foam may seem at first glance, it still remains a complex system with a gas dispersed phase and a liquid / solid dispersion medium. If we talk about the most common foam, consisting of gas bubbles and liquid films, then such a structure is considered a nonequilibrium system. Foam can also be called a polydisperse system due to the fact that the composite bubbles can be of completely different sizes. In addition, the foam is very unstable and therefore very short-lived system due to the fact that the density of the liquid is hundreds or even thousands of times higher than the density of the gas.
Despite this, foams are quite common in human life and are used in various fields. They are present in everyday life (whipped cream, shaving foam, etc.), in biotechnology (foam in bioreactors), in chemical technology (foam flotation), and even in pharmacology. If we study the mechanism behind the process of destruction of the foam, then we can make it more durable, as the researchers themselves say.
In their work, they point to three main processes that regulate the dynamics of the foam: enlargement, drainage and destruction. Enlargement is the process of reducing the number of bubbles, but increasing their size, which is caused by different pressures between the bubbles. Draining is the process of thinning the films, that is, the walls of the bubbles, due to the flow of water under the influence of gravity.
These two stages of foam life have already been studied quite well, as, in general, the process of destruction. Previous studies have shown that bubble destruction occurs when the lower limit of coalescence * in the volume fraction of liquid is reached.
Coalescence * - the fusion of particles within a mobile medium (for example, bubbles in the foam).A relationship was also established between the destruction of the bubble and the rearrangement of T1 * by introducing additional air into the system.
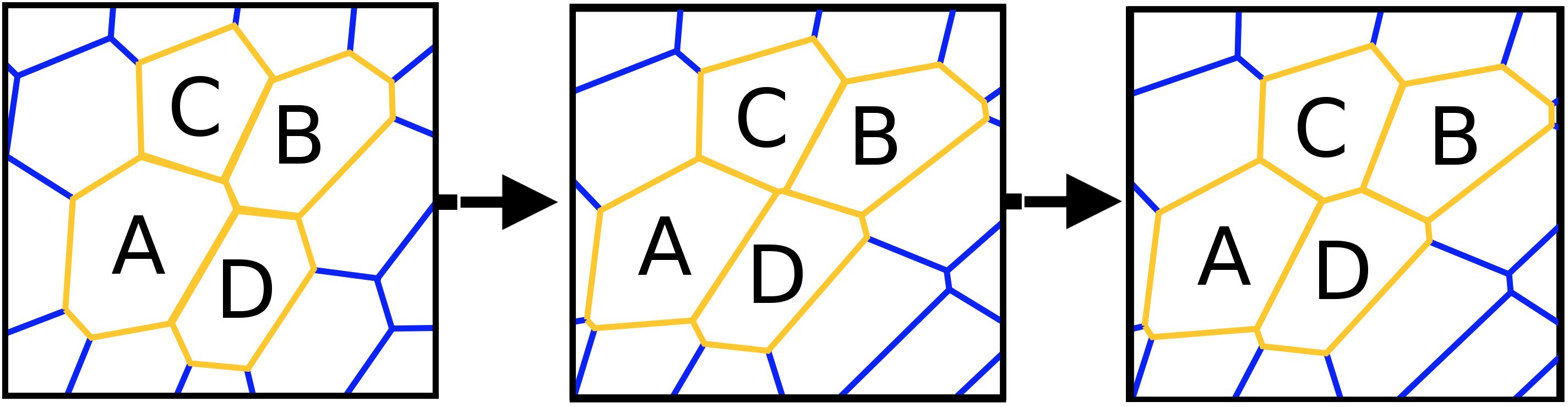
The T1 * process or T1 * rearrangement is the process of changing the shape of cellular materials (foam, biological tissues, etc.), consisting of drops, bubbles, cells.At the moment of destruction of the bubble, one could hear a slight “pop” (gas release). By measuring and analyzing the acoustic data at the time of the destruction of the foam, scientists came to the conclusion that this process occurs due to collective collapse of the bubbles (CCP), in other words, cascade collapse.
At the beginning of the process, there are 4 objects (A, B, C and D). A and B are in contact, C and D are located on both sides of AB, that is, they are not in contact with each other. The breaking of the connection between A and B with the subsequent establishment of communication between C and D - this is the process T1.
But KKP is just the tip of the iceberg, and the mechanism itself, which launches it into action, is unknown. This is how to understand that the chain of dominoes is destroyed, because the dominoes fall on each other, but do not know which one was the first and that it was knocked over (a rough, but understandable allegory).
In this work, scientists used quasi-two-dimensional foam as an “experimental”, on the edge of which 1 bubble bursts. Scientists observed the cascade of destruction of the entire foam using a high-speed camera, after which they analyzed the records. Two mechanisms of collapse propagation have been established: propagation and penetration.
As φ (volume fraction) increases , it becomes more and more difficult for liquid droplets to penetrate liquid films, which causes the drop to bounce off the films due to their elasticity or the absorption of droplets by the film. More about this in the results of observations.
Research results
The process of CCP (collective bubble collapse) was observed at different values of φ . So, for example, image 1a shows the CCP from time t = 0 ms to 3.12 ms for φ = 0.0099.
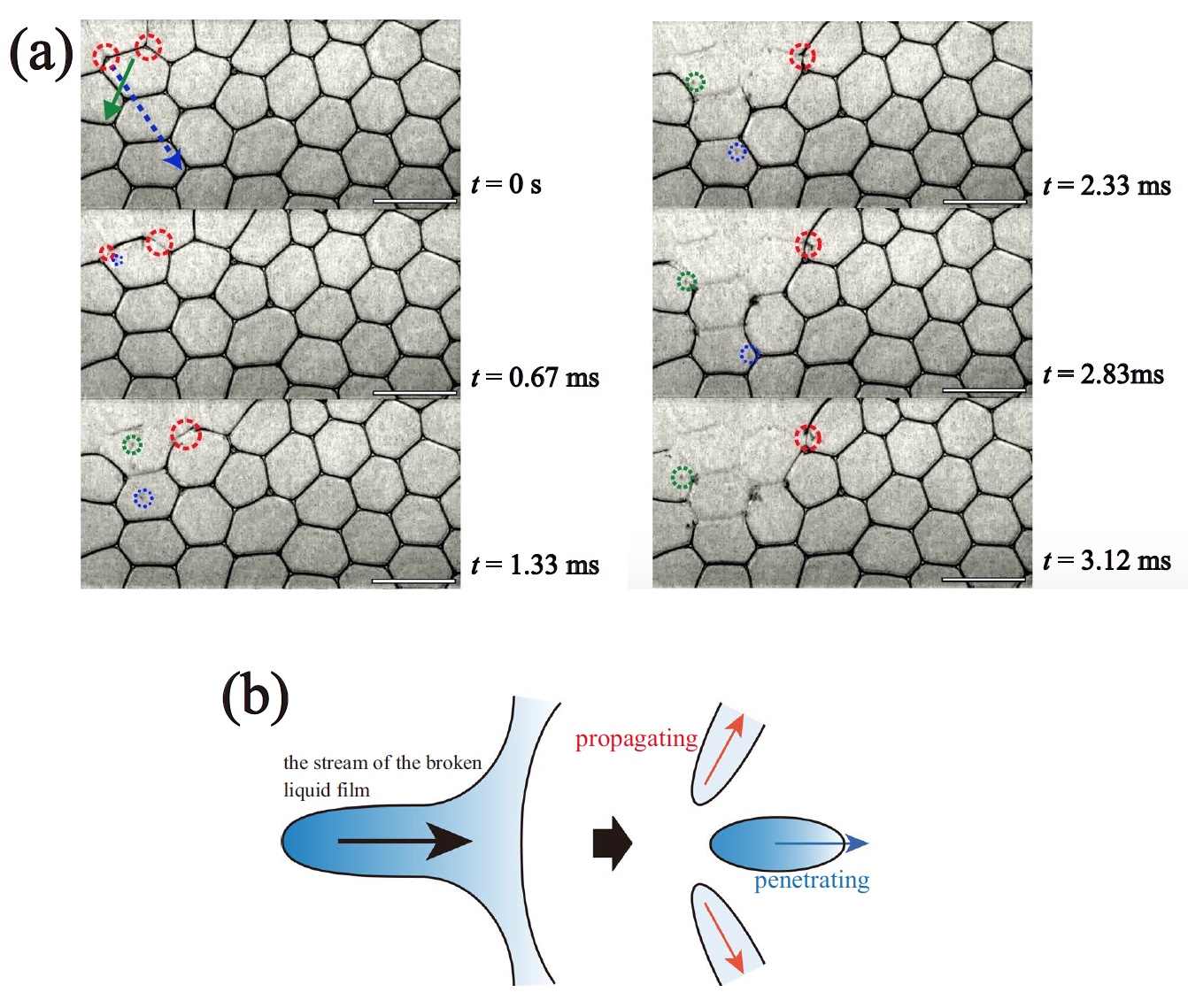
Image No. 1

The KKP process ( t = 0 ... 3.12 ms, φ = 0.0099).
A capillary glass needle was used to puncture the bubble. The collapse of the bubbles at the beginning of the CCP process is observed along the outer edges of the foam, which scientists decided to call the surface effect. Following this, two CCP processes begin inside the foam itself, so to speak in its total volume: spread and penetration.
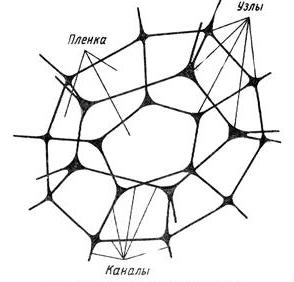
When the liquid film is torn, it is quickly absorbed by the Plateau channel.
According to the Plateau law, the edges of the bubble in the foam are channels filled with a dispersion medium. Only three films located at angles of 120 ° can converge in one channel.Due to the strong absorption effect, the next liquid film converging in the same Plateau channel also breaks (red circle on 1a ). This sequential (cascade) process of destruction of foam bubbles is one of the processes of CCP - propagation ( 1b ).
The structure of the cell (bubble) of the foam.
At the same time, a drop of liquid (blue and green circles on 1a ) is released at the time of absorption of the bursting film by the Plateau channel . Drops fall into the foam into the film removed by the liquid (arrows on 1a ). The velocity of these drops (V d ) was approximately 3 m / s. This CCP process, called penetration, leads to the destruction of the removed films ( 1b ).
Complete collapse occurs during the multifaceted destruction of the bubble films through both CCP variants.
If φ is increased to ≥ 0.015, then the probability of a drop of liquid at the time of destruction of the bubble film is greatly reduced. And the speed of the droplets that have arisen also becomes less, which makes it more difficult for the droplets to penetrate the removed films. Instead of penetration, a drop bounces.

Drop rebound (instead of penetration) at φ ≥ 0.015.

Image No. 2
The image above shows how a drop bounces off the films within 30 ms (the dotted line is the trajectory of the drop).
By measuring the droplet velocity (V d ) after each rebound, we can plot the dependence of V dof the number of strokes (n i ).
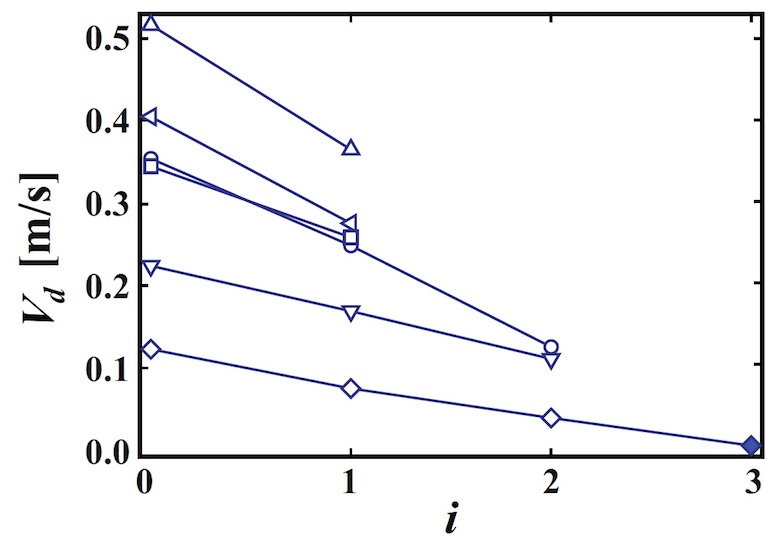
Image 3: Speed versus the number of bounces.
As expected, the droplet velocity decreases with increasing number of bounces. In this case, it is possible to determine the film recovery coefficient as e = | V d (i + 1) | / | V d (i) |, where | V d (i) | - droplet velocity after the i-th rebound. Using observational data, it was found that e = 0.50 ~ 0.74. After the ith rebound, the drop is absorbed by the liquid film.
With a further increase in φ (> 0.022), the film is successfully absorbed by the Plateau channel, but liquid drops do not arise. Bubbles along the edge of the foam burst from the surface effect and at higher ratesφ , but the number of such bubbles is greatly reduced, and the collapse process itself quickly stops. In other words, the CCP process does not occur.
Next, scientists investigated the dependence of the number of bursting bubbles on the index φ. The set of collapsing bubbles (N total ) consists of those that burst at the edge of the foam due to the surface effect, and those that collapse due to penetration and propagation.
Also in the calculations, the indicator N inner is used - the number of collapsing bubbles in the foam volume minus the outer edges. The destruction of the bubbles was counted from the first to the last bubble, which took about 0.04 seconds.
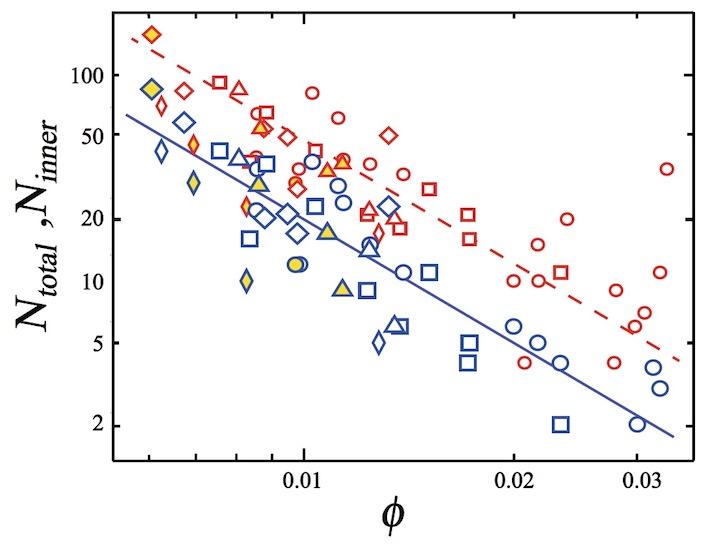
Image No. 4
The image above shows the values of N total(red) and N inner (blue) relative to φ . Triangles, circles and squares correspond to N total or N inner at N f ~ 200 for glycerol concentrations of 9.4%, 17.8% and 29%, respectively (N f is the total number of bubbles in the foam).
As we can see from the graph, the value of N total and N inner decreases with increasing φ . Applying the power law * , scientists found that N inner ∝ φ −γ e at γ e = 2.3 ± 0.36.
The power law * is the functional dependence of two quantities when a change in one leads to a proportional change in the other.It was also found that the indicators N total and N inner do not depend on the concentration of glycerol, if it is below 29%. If the concentration increases to 40%, then it becomes more difficult to pierce the bubble, and the CCP process does not occur.
The study of collapsing bubbles in the case of a larger foam (N f ~ 500) showed that their number does not depend on the total number of bubbles (diamonds in the graph above), that is, N total and N inner are independent of N f .
As we recall, a glass needle was used to puncture. It was coated with silicone grease to improve piercing. Scientists have tested how this affects the value of N total and N innerby making punctures without lubrication. Thus, the CCP process arose spontaneously. However, as one would expect, the use of the lubricant did not affect the number of collapsing bubbles and the CCC process as a whole.
If φ is small, then the shape of each bubble is anisotropic, and distorted bubbles form a kind of chain. Bubbles of anisotropic shape and / or large size have a large excess surface energy, therefore, they are more easily destroyed.
Given this, scientists decided to check the relationship between CCP and the shape of the bubbles. For this, the parameter λ i was used as a characteristic of the anisotropy of bubble i . λ i is determined by the following formula:
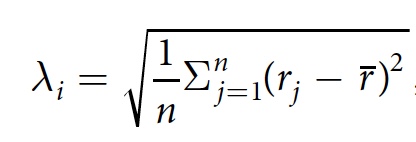
where jIs the pixel at the edge of the bubble, n is the total number of pixels j , r j is the distance between the center of bubble i and pixel j , r is the average distance r j .
λ i will be equal to 0 if the bubble i is round. If it is anisotropic, then λ i > 0.
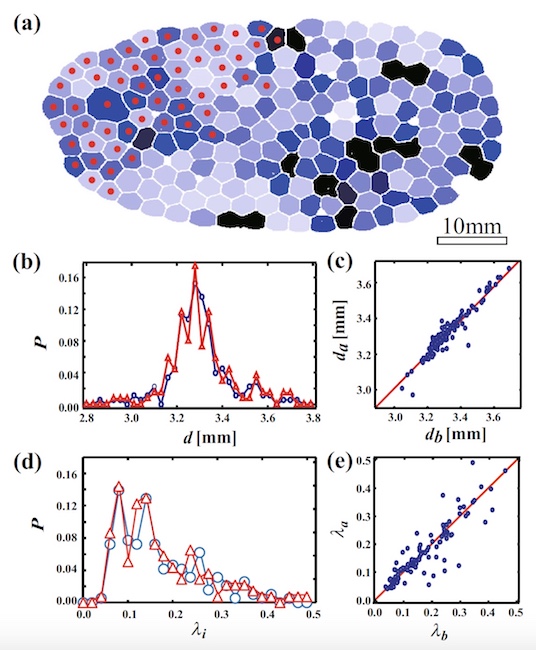
Image No. 5
Image 5a shows foam at φ = 0.0086 before the beginning of the CCP process. Bubbles are colored from black (λ i more) to white (λ i less). Red dots indicate that the bubbles collapsed during the CCP.
Scientists have found that the bubbles on the left are evenly destroyed. Graph 5b shows the probability distribution as a function of the mean bubble diameter i (d i ) before and after the PEC of all bubbles. The diameter (d i ) was calculated by averaging the distance between the center and the interface of the bubble. As we can see, the form of the probability distribution on the graph after CCP is the same as before CCP.
On the graph 5c, the ratio of the diameter after the CCP (d a ) and to the CCP (d b ). Obviously, d a = d b , that is, the diameter of the bubbles did not change during the cascade collapse. 5d
chartshows λ i (characteristic of bubble anisotropy) before and after the collapse. This indicator also did not change, despite the cascade collapse (λ a (before CKP) = λ b (after CKP); 5e ).
All these observations indicate that the shape of the bubbles does not change due to the cascade destruction of the foam, and also that the shape does not affect this process.
The next probable factor affecting the process of foam destruction is the mechanical effect of a torn bubble film on its neighbors. To determine the influence of this factor, scientists measured the rate of film rupture at a concentration of glycerol of 17.8% according to the formula V = l / t , where l is the length of the film, andt is the time required to absorb the film, from start to finish.
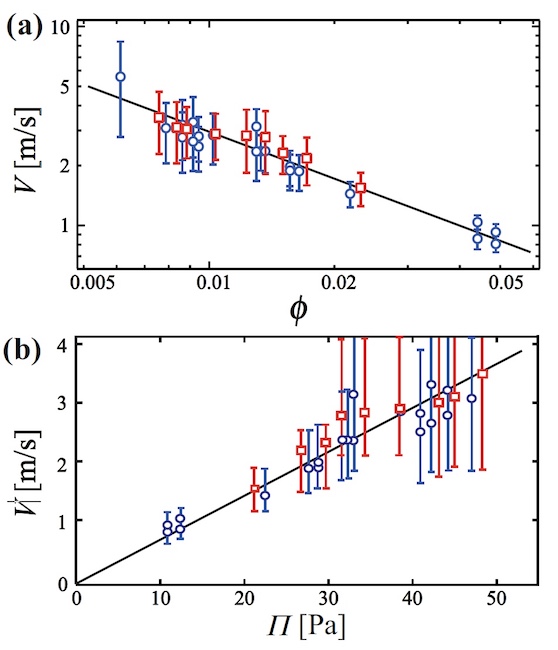
Image No. 6
Graph 6a shows the dependence of V on φ in the form of a logarithmic graph. Calculations show that V ~ 10 m / s at a glycerol concentration of 17.8% (circles on the graph). In the case of a concentration of glycerol of 29%, the rate practically did not change (squares on the graph).
With increasing φ, the velocity decreases, due to which the films formed during rupture bounce off other channels and, as a result, are absorbed by them.
The relationship between velocity and osmotic pressure ( 6b ) has also been studied .
The pressure formula for two-dimensional foam is as follows:
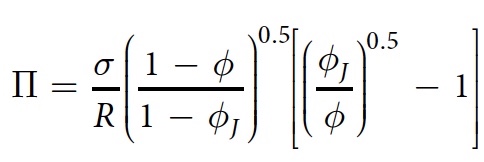
where σIs the surface tension, R is the average radius of the bubble, φ J is the jamming point of 0.16 in two dimensions.
In their work, scientists used the following indicators: σ = 37 mN / m and R = 1.7 mm.
If we assume that the film layer thickness is 1 μm, then a proportional dependence of V on Π ( 6b ) is visible . Therefore, the driving force of absorption is the negative pressure in the film.
Finally, scientists conducted an analysis of the ratio of N inner and speed V (image below).
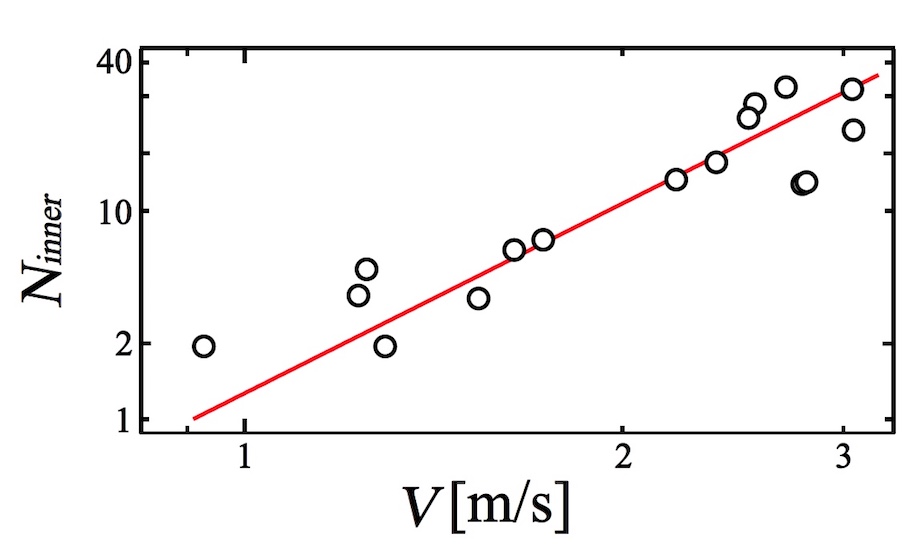
Image No. 7
Scientists have found that the indicator N innergreatly increases with increasing film flow rate. Thus, we can conclude that the penetration process is a decisive element in the process of general foam collapse.
For a more detailed acquaintance with the nuances of the study, I recommend that you look into the report of scientists .
Epilogue
In this work, scientists were able to find out that at the time of the destruction of the foam, the main role is played by two processes - absorption and distribution. In addition, scientists found that an increase in the proportion of liquid, the speed of the drops that occur during the destruction of the bubble film decreases. Therefore, it is more difficult to destroy all the foam. Instead of absorbing a drop by another distant film, a multiple drop of the drop occurs, and only then absorption.
Scientists intend to further study the foam in order to understand its strengths and weaknesses. In their opinion, this work will improve the foam, making it more durable and stable. And such advantages can be useful both in everyday life and in laboratories involved in the production and study of various biological and chemical substances, materials and other things.
Who would have thought that in the 21st century, scientists would actually study beer foam, looking for ways to make it stronger. But, no matter how strange it may sound, any knowledge is important, any knowledge is needed. Understanding the world around us and everything that fills it allows us to better use what was invented or discovered long ago, or to improve it in accordance with the constantly changing conditions of our lives.
Thank you for your attention, remain curious and have a good working week, guys! :)
Thank you for staying with us. Do you like our articles? Want to see more interesting materials? Support us by placing an order or recommending it to your friends, a 30% discount for Habr users on a unique analogue of entry-level servers that was invented by us for you:The whole truth about VPS (KVM) E5-2650 v4 (6 Cores) 10GB DDR4 240GB SSD 1Gbps from $ 20 or how to divide the server correctly? (options are available with RAID1 and RAID10, up to 24 cores and up to 40GB DDR4).
Dell R730xd 2 times cheaper? Only we have 2 x Intel TetraDeca-Core Xeon 2x E5-2697v3 2.6GHz 14C 64GB DDR4 4x960GB SSD 1Gbps 100 TV from $ 199 in the Netherlands! Dell R420 - 2x E5-2430 2.2Ghz 6C 128GB DDR3 2x960GB SSD 1Gbps 100TB - from $ 99! Read about How to Build Infrastructure Bldg. class using Dell R730xd E5-2650 v4 servers costing 9,000 euros for a penny?