About yellow phosphorus and the panicky nature of man

Hi% username%.
As promised - here's an article about the yellow phosphorus and how it burned nicely under Lviv in Ukraine relatively recently.
Yes, I know - Google gives a lot of information about this accident. Unfortunately, most of what he gives out is not true, or, as eyewitnesses say, nonsense.
Let's figure it out!
Well, at first - nobody’s favorite equipment, but it, by the way, is very important!
As boring Wikipedia says, phosphorus is one of the most common elements of the earth's crust: its content is 0.08-0.09% of its mass. In a free state does not occur due to high chemical activity. It forms about 190 minerals, the most important of which are apatite Ca 5 (PO 4 ) 3(F, Cl, OH), phosphorite Ca 3 (PO 4 ) 2 and others. Phosphorus is part of the most important biological compounds - phospholipids. Contained in animal tissues, part of proteins and other essential organic compounds (ATP, DNA), is an element of life. Remember this,% username%, and we will move on.
Pure phosphorus is white, red, black and metallic. This is called allotropic modifications - the weak sex is very well understood in them, because it can distinguish a diamond from graphite by touch - and these are also allotropic modifications, only for carbon. In general, phosphorus is the same.
The hero of our story - yellow phosphorus - is actually raw white. Very often, "untreated" - this means an admixture of red phosphorus, and not some terrible foreign elements there.
Yellow phosphorus (however, like white) is the real hell: highly toxic (MPC in atmospheric air 0.0005 mg / m³), flammable crystalline substance from light yellow to dark brown in color. Specific gravity 1.83 g / cm³, melts at +43.1 ° C, boils at +280 ° C. It does not dissolve in water; it easily oxidizes and self-ignites in air. It burns with a dazzling bright green flame with the emission of dense white smoke - small particles of tetraphosphorus decoxide P 4 O 10 . This is again boring Wikipedia, but I ask,% username%, - remember this information.
Now we will understand.
Well, firstly, despite the toxicity of phosphorus, it is extremely difficult to poison them for a very simple reason: it spontaneously ignites in air. Very fast. And it burns, as has already been said, with a
Although, maybe you can argue, they write: the lethal dose of yellow phosphorus for humans is 0.05-0.15 grams, it dissolves well in body fluids and, when ingested, is rapidly absorbed (by the way, red phosphorus is insoluble and therefore relatively low-toxic). Acute poisoning occurs when the vapors of yellow phosphorus are inhaled and / or when they enter the gastrointestinal tract. Poisoning is characterized by abdominal pain, vomiting, beautiful vomit glowing in the dark, emitting the smell of garlic, diarrhea. Another symptom of acute yellow phosphorus poisoning is heart failure.
After reading this, for some reason I remembered about phosphine poisoning (the symptoms are very similar) and thought hard - but not about the existence of yellow phosphorus vapors, but about the adequacy of the individual who saw a piece of something that was smoking, glowing in the dark - and immediately it ate. Well then.
By the way, to get a solution of phosphorus in water 3 mg / l - and this is a saturated solution, it does not dissolve anymore - you need to shake a piece of phosphorus in water for a week. Well, I didn’t come up with it, GOST 32459-2013 says so - and this is not all kinds of Internet for you!
In general, in my opinion - the toxicity of phosphorus is very exaggerated. But he has other nuances. About them - below.
Phosphorus burns, as the experts who work with it like to say, according to the rule of the gimlet: that is, a burning piece eats into the surface on which it burns. To the table. In the metal. Into the boot. In the hand. The reason is simple: the combustion product, phosphorus oxide, is essentially an acid oxide that immediately draws water to form phosphoric acid. Phosphoric acid, although not as tidy as sulfuric or hydrofluoric acid, loves to eat no less - and therefore it corrodes everything. By the way, it is sometimes added to the toilet cleaning liquid. A nice combination of high-temperature combustion (up to 1300 ° C) and hot acid gives extra holes to your table, and if you're not lucky, your body. And yes,% username% is very painful.
I have argued many times and will continue to say that there is no greater enemy to man than himself: of course, the properties of yellow phosphorus did not go unnoticed - and good people came up with the idea to add it to incendiary ammunition, because it is very convenient when something suddenly lights up in the air !
It looks very beautiful - you can admire









But people after such attacks do not look very pretty - so do not look better

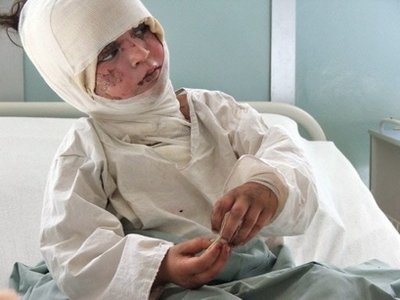
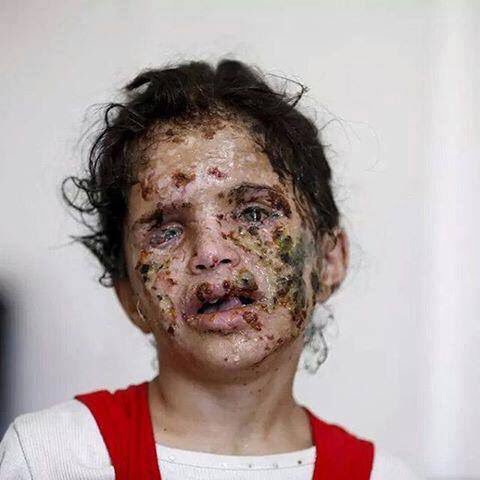
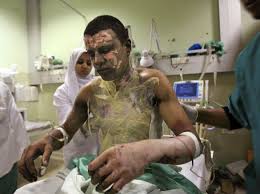





Since all this is very charming, the development, testing, transportation, trade, use and disposal of phosphorus munitions are carried out taking into account a number of international agreements and treaties, including:
- St. Petersburg Declaration on the Abolition of the Use of Explosive and Incendiary Bullets of 1868.
- Additional 1977 protocols to the 1949 Geneva Convention for the Protection of Victims of War prohibiting the use of white phosphorus ammunition if civilians endanger it. The United States and Israel did not sign them, by the way.
- In accordance with the Third Protocol to the UN Convention on specific types of weapons of 1980, incendiary weapons should not be used against civilians, and, in addition, it should not be used against military installations located in the concentration zone of civilians.
In general, there are a lot of papers, but they have a status close to the toilet, because these ammunition is used all the time - Palestine and Donbass will confirm.
Since phosphorus reacts with water only at temperatures above 500 degrees Celsius, large quantities of water are used to extinguish phosphorus (to lower the temperature of the source of ignition and solidify phosphorus) or a solution of copper sulfate (copper sulfate), after quenching, phosphorus is poured wet sand. To protect against spontaneous combustion, yellow phosphorus is stored and transported under a layer of water (a solution of calcium chloride, to be precise, but the water will also come off). This is also important!
Who produces phosphorus? And here,% username%, some will feel proud: the main supplier of phosphorus, food phosphoric acid, sodium hexaphosphate and tripolyphosphate is proud Kazakhstan!
In fact, since the days of the USSR, the company Kazphosphate was built in the glorious city of Dzhambul (yes, the name of that same Dzhambul Dzhabaev). Then Jambul was renamed to Taraz - well, we won’t discuss expediency, the Kazakhs know better - but the enterprise remains. The presence of a raw material base and capacity, as well as the extremely low cost of labor (and there is nowhere to work in Taraz / Dzhambul, in fact) caused yellow phosphorus to be produced right here.
When I was at this enterprise - it’s good there! South Kazakhstan, 300 km to Uzbekistan - warm! Birds are singing! Everything is green! Mountains are on the horizon! Beauty!
By the way, the plant of Kazphosphate does not violate this idyll in any way: all in greenery, flowers, on the slope of a small mountain.
There really is good



The reason for beauty is simple - phosphorus-containing substances, which are actually fertilizers, are raw materials, products and industrial wastes. That's all and grows, blooms.
By the way, the highest authorities of the plant really do not like dandelions. No one knows why. That is why, before the visit of the highest authorities, workers are given a clean-up day for the dandelion shedding. Well, how it is - to fight with dandelions - everyone knows in the country / gardens, in the framework of phosphorus lawlessness this is completely pointless: enough for a day, a maximum of two. But leadership is it.
I was especially impressed with the work of the laboratory of the enterprise. There are really big smart women sitting there. And so you understand,% username%, a bit of facts.
In yellow phosphorus, it is very important to control impurities - especially arsenic, antimony, selenium, nickel, copper, zinc, aluminum, cadmium, chromium, mercury, lead, iron. In order to control all this - phosphorus must be dissolved, and at the same time everything that is controlled should not fly away.
Task number one: how to weigh what lights up in the air? They do this: hammer a phosphorus ingot under a layer of water, take larger pieces - small ones flare up too quickly - and transfer to a glass of water. Then they weigh another glass with water, take phosphorus from the first, wipe it with alcohol, wait until it dries - and throw it into a weighted glass with water. The weight difference determines the mass of phosphorus.
Since it can catch fire - a solution of copper sulfate stands next to it - if it ignites, then it is thrown into it.
Then phosphorus is dissolved. It is soluble in nitric acid saturated with bromine vapor - a very sweet and fragrant thing, I recommend it on the farm (no). It is necessary to throw phosphorus into this mixture, then heat it a little, and when the reaction proceeds, transfer it to the trough with cold water, because the heating is colossal. And interfere, interfere, interfere - if not interfere, then the pieces will simply jump out of the seething soup - the results will be inaccurate! They interfere with the hand, there are two mittens on it: rubber from acid - and felt from temperature (just rubber melts, and just felt melts - it does not save acid drops. True, if phosphorus gets into it, both will not save.
A fascinating sight of the dissolution of yellow phosphorus









At the same time, nitrogen oxides and bromine fly - this is a note. The girls are afraid of these particular red tails and pieces of phosphorus, which can get on clothes or mitt. Poisoning by “vapors” or “solutions” of phosphorus is not recalled.
By the way, the salary of the girls who do this is no higher than $ 200 (and the answer is simple: there is nowhere else to work in Taraz, I already said that). So next time,% username%, when you whine about low salaries and the harmfulness of work - remember Kazphosphate!
Well, now, when the basic knowledge has been accumulated, we are moving on to the accident in Lviv.
Since phosphorus is in demand in Europe, Kazphosphate is actively exporting products through Czech partners. She rides in tanks filled with water, and it is clear that by rail.
On Monday, July 16, 2007, at 16:55 in the Busk district of the Lviv region of Ukraine, on the Krasnoye-Ozhidiv stage, 15 tanks with yellow phosphorus of freight train No. 2005 rolled over and rolled over. In total, there were 58 cars. Tanks followed from the Kazakh station Asa (Taraz, Kazakhstan) to the station Oklesa (Republic of Poland). A leak of phosphorus from one tank provoked spontaneous combustion of six other tanks.
It looked epic















And then - a mixture of panic, bloated media, a lack of experience with yellow phosphorus and complete ignorance of chemistry.
During a fire extinguishing, a cloud of combustion products formed with a zone of destruction of about 90 square kilometers. In this zone there were 14 settlements of the Busk region, where 11 thousand people live, as well as certain territories of the Radekhiv and Brodiv districts of the region. The Ministry of Emergencies of Ukraine invited residents of nearby villages to evacuate and sent them about ten buses, but many people refused to leave their homes. The Lviv authorities assured that they would not forcibly evacuate anyone, although they warned about the unpredictability of the consequences of the accident. In total, about 800 residents were temporarily resettled out of 6 settlements of the Busk district per night.
By Tuesday, there were 20 people injured (6 specialists of the Ministry of Emergencies, 2 representatives of the Ministry of Internal Affairs, 2 railroad workers and 10 people from the local population), of which 13 in serious and moderate condition were hospitalized in the military medical clinical center of the Western Operational Command in Lviv. Seven hospitalized - employees of the Ministry of Emergency Situations, two - employees of the State traffic inspectorate, four - local residents.
At the same time, a fierce and wildest howl rose in the media. Some of the pearls:
- My beloved, about coughing cats and dogs and the second Chernobyl
- Washington Post
- Vesti.RU
- Bbc news
- Chron
- ... and another over 9000, which Google finds with the right question
Reading all this makes me sad. Because it shows absolute ignorance of chemistry in the mass. And also - how easy it is to manipulate the uneducated mass (by the way,% username%, and you knew that slave owners in the United States firmly believed that slaves should be illiterate so that they could not fake vacation certificates, other papers, correspond with other settlements, and coordinate uprisings etc. - little has changed).
More or less objective events in the chronology are shown here (carefully - Ukrainian, if you don’t know shame - Google Translate):
What can be understood from this chronology?
- No one knew anything.
- Everyone wanted to PR.
- Firefighters / MES were scared.
- The military, too.
- Among the locals was a complete mess.
- Until July 18, representatives of Kazphosphate arrived - no one understood what to do.
- No one wanted to pay for anything.
After talking with some employees of Kazphosphate directly involved in the aftermath of the accident, I can say the following.
There was no explosion / spontaneous combustion / undermining of phosphorus - he rode quietly under his own water. Yes, and yellow phosphorus itself does not know how to explode! But there was damage to the railroad track, because of which the tanks went off the rail. When the tanks hit, a crack formed, water flowed out through it - well, phosphorus was safely ignited.
The temperature and combustion characteristics completely destroyed the tank.
- White smoke is quite explainable - this is a pair of phosphoric acid, but not phosphorus. If you breathe them, yes, a strong cough will begin, and in general it is not particularly useful. However, it is not deadly harmful. Most of the injuries of the local population are due to the fact that people ran to collect interesting smoking pieces in water bottles, but did not immediately put on the cordon - everyone was afraid.
- The fear of firefighters that supposedly "this rubbish from the water is burning!" Is due to the fact that a powerful stream of water broke phosphorus into smaller pieces - well, they scattered and caught fire. It was necessary either with a weak stream, or with foam, which was subsequently done.
- By the way, when everything was extinguished and there were only pieces inside the tank, the Kazakhs extinguished it. Well, as they put out, they collected and threw into buckets of water for the most part. One of them is the chief technologist of the plant, a heavy smoker. So - he put out and smoked. Even the pictures of the “crazy Kazakh, who also smokes in a terrible chemical fire, have dispersed in some places!” But what?
- There was no ecological catastrophe and the “second Chernobyl” and could not be - in fact, nature received a dose of phosphate fertilizers.
- The only person who behaved appropriately listened to the Kazakhs and did what was right - Vladimir Antonets, First Deputy Minister of Emergencies. Probably because the Colonel General with a bunch of awards.
After it became clear that there was no sensation: there was no terrorist attack, no threat of environmental disaster — no one died
- Inadequate condition of tracks on this railway line.
- Violation of safety rules by employees of the locomotive crew.
- Negligence (instructions on the temperature regime of transportation of especially dangerous goods were ignored).
- Inadequate technical condition of tanks.
In fact, the most true of this is the first. The rest was added so as not to pay the Kazakhs for the loss of cargo. Well, it seems like the insurance compensated.
So everyone remained with their own.
Morality,% username%: learn chemistry . She is everywhere. She will help you live and survive, and understand something for yourself.
And finally ...
Not all chemical compounds are harmful. Without hydrogen and oxygen, for example, it would be impossible to get water, the main component of beer.- Dave Barry, never a chemist
