Anti-Aging News
Increasing the time between meals and calorie restriction prolongs life
In September 2018, Cell Metabolism published a study by American gerontologists from the National Institute on Aging (NIH) about the effect of time between meals on life expectancy. Experimental mice were divided into two groups. The first group had a diet with natural nutrition (a reduced amount of refined carbohydrates and fats, and an increased content of proteins and fiber). The other group of rodents, on the contrary, was planted on an “unhealthy” diet - with an increased amount of simple carbohydrates and fats, and a reduced one - of proteins and fiber. In addition, the mice in each group were divided into three subgroups, based on how often they have access to food. The first subgroup of mice had access to food around the clock. The second subgroup of mice was given food once a day, and the number of calories in their portions was the same as in the mice of the first subgroup, i.e. not trimmed. The third subgroup was fed a diet reduced by 30% of calories.
The second and third subgroups, according to the observations of scientists, had a stronger appetite and quickly ate the food brought, which led to longer daily periods of fasting for both groups.

Figure from the article.
The first subgroup of mice ( ad libitum ) - fed to the fill, who had access to food 24 hours a day.
The second subgroup of mice ( meal-fed ) - received food once a day, without cutting calories.
The third subgroup of mice ( CR, calorie restriction ) - received a diet reduced in calories by 30%.
Throughout the study, scientists monitored the metabolic health of mice until natural death, and then studied them posthumously. It was found that mice from two subgroups, which were fed once a day and limited in calories, improved overall health. As evidenced by the delay in general age-related damage to the liver and other organs, as well as an increase in life expectancy. Mice that were limited in calories also showed a significant improvement in blood glucose and insulin levels compared with other groups. In general, mice had such indicators of life expectancy. Mice that were fed normally, without restrictions - lived 104-110 weeks, with one-time meals per day - 115-117 weeks, with calorie restriction - 132-135 weeks.
As the autopsy showed, all three groups of mice had the same pathology, but different periods of their development. The main non-tumor tissue damage was amyloidosis, and the accumulations of amyloid were more extensive in long-lived mice fed with calorie restriction. Scientists explain this fact by the longer lifespan of these mice.
It is noteworthy that according to the observations of the researchers, the dietary composition did not have a significant impact on the life expectancy in the subgroups with a single meal and calorie restriction. Scientists attributed this to the fact that large intervals between meals allowed the body to more effectively use recovery mechanisms: “Increasing daily fasting periods without reducing calories and regardless of the type of diet consumed led to an overall improvement in health and survival in male mice. Perhaps this extended daily period of time on an empty stomach allows for the restoration and maintenance of mechanisms that would be absent with prolonged exposure to food. ” [3]
Mediterranean diet reduces the risk of stroke in women
The Mediterranean diet can reduce the risk of stroke in women older than 40 years, but not in men, such data were obtained in a new study conducted by British cardiologists.
An article published in the Journal of the American Heart Association in September 2018 describes that a diet high in fish, fruits, vegetables, nuts and beans, as well as fewer meat and dairy products, reduces the risk of stroke among women who are at high risk of heart disease. -vascular diseases. This study, according to scientists, is one of the largest and longest, aimed at assessing the potential benefits of a Mediterranean-style diet in reducing the risk of stroke. It shows that such a diet can be especially effective in women over 40, regardless of the state of menopause or hormone replacement therapy.
Scientists from universities in East Anglia, Aberdeen, and Cambridge, as part of an EPIC study in Norfolk, which lasted 17 years, studied the diets of 23,232 participants and compared the risk of stroke depending on the diet of the participants.
The participants who most consistently adhered to the Mediterranean diet, 22% of women and 6% of men had a reduced risk of stroke.
“This study shows us that a Mediterranean-style diet rich in fish, fruits and nuts, vegetables and beans, and with less meat and dairy products, can reduce the risk of stroke in women over 40 years old. But a healthy balanced diet is important for everyone, both young and old. It is not clear why we found differences between women and men, it is possible that the components of the diet can affect men differently than women. We also know that different stroke subtypes may differ between the sexes. Our study was too small to test this, but both possibilities deserve further study in the future, ”the researchers write.
The American Heart Association, in its recommendations, also focuses on a diet similar to the Mediterranean diet: "The American Heart Association recommends a healthy lifestyle and a diet that includes many fruits and vegetables, whole grains, low-fat dairy products, fish, poultry, beans, not tropical vegetable oils and nuts, as well as limiting saturated fats, trans fats, sodium, red meat, sweets and sugar-containing beverages; these dietary ingredients reduce the risk factors for heart disease and stroke ”[4]
MiRNA-141-3p, signal molecule SDF-1 and age-related bone tissue dysfunction
In August 2018, the Journal of Gerontology published an article about the relationship of miR-141-3p, the SDF-1 signaling molecule (Stromal cell-derived factor-1), and bone aging.
According to the International Osteoporosis Foundation, about 30 percent of postmenopausal women in the United States and Europe have osteoporosis. At least 40 percent of these women and about 30 percent of men of the same age have an increased risk of fractures due to the fragility of their bones for others.
Mesenchymal stem cells can differentiate into the main components of our skeleton: osteoblasts forming bone cells, osteocytes, cartilage cells, chondrocytes; as well as adipocytes in fat cells. SDF-1 is a key signaling molecule that regulates the differentiation of stem cells into these cells, studies have shown. SDF-1 also has many other functions, including helping mesenchymal stem cells to get to the right place during bone formation or repair. In addition, SDF-1 acts as an antioxidant, protecting cells from the damaging effects of oxidative stress.
The great importance of SDF-1 in maintaining normal bone health, and the fact that the level of this molecule decreased with age, made scientists interested in how it is regulated. Scientists have suggested that reducing SDF-1 levels, at least in one of the ways, depends on miR-141-3p.
Previous studies have shown that the role of miR-141-3p may be negative. It was found that this miRNA inhibits the activity of the vitamin C transporter, which did not allow the vitamin to enter the cells. Vitamin C is also important for bone health, and without sufficient transporters, vitamin instead begins to accumulate outside the cell, where it causes oxidative stress. Animal studies have shown that oxidative stress in mesenchymal stem cells reduces SDF-1 levels. collecting all the available data together, the researchers suggested and then found that higher oxidative stress increases miRNA-141-3p expression, which, in turn, reduces the levels of SDF-1.
In both mouse and human mesenchymal stem cells, it was found that microRNA-141-3p levels are lower in young cells. And in old cells, the level of this miRNA increased 3 or more times. For levels of the SDF-1 molecule, a fundamentally opposite phenomenon was found - a multiple decrease with age. Adding microRNA-141 to stem cells resulted in lower levels of SDF-1. The consequences of this include another age shift: the production of more bone osteoclasts, to the detriment of osteoblasts. It was also noted that mesenchymal stem cells more differentiated into fat cells, which is also usually associated with age.
As part of a comprehensive testing of their hypothesis, scientists first added miR-141-3p to cells, and the observed function of bone tissue deteriorated, then used an inhibitor of this micro RNA, and improvement was observed.
According to researchers, clinical drugs, such as the drug used to inhibit microRNA-141-3p in their work, could one day become an effective way to help mesenchymal stem cells to remain functional, regardless of age and other conditions: “The inhibitor normalizes the function bones. We believe that a clinical-level inhibitor can help us do the same in humans. ”
At the next stage, scientists plan to switch to animal models and look at many other factors involved in the aging of bone tissue. And also find out whether higher levels of physical activity, which tend to decrease with age, can help restore a healthier balance of miR-141-3p and SDF-1. It is also planned to investigate other members of the microRNA-141 family for their participation in the aging process [5].
How do “bad” bacteria conquer the intestines
In September 2018, an article by English scientists was published in the journal PLOS Pathogens, which described the mechanism of colonization of intestinal pathogens by pathogenic bacteria after taking antibiotics.
It is already known that antibiotics violate the natural protective intestinal flora, making people susceptible to the pagonne bacterium Clostridium difficile. Infection with this pathogen (CDI) causes most diarrheal outbreaks associated with antibiotic therapy, and can lead to potentially life-threatening diseases and complications. C. difficile can cause dangerous weight loss and attenuation over a short period of time. Currently, there is a great need to better understand how C. difficile can affect the intestinal microbiota and disrupt intestinal homeostasis. Reproduction of this bacterium occurs in conditions when antibiotics kill other pathogens.
C. difficile is a gram-positive spore-forming anaerobe that is in harsh conditions for its life for long periods of time. Antibiotic treatment directed against this bacterium does not give the desired result. 35% of patients are again susceptible to the recurrence of C. difficile infection. These facts led scientists to look more closely at this bacterium in order to understand what makes it so weakly vulnerable.
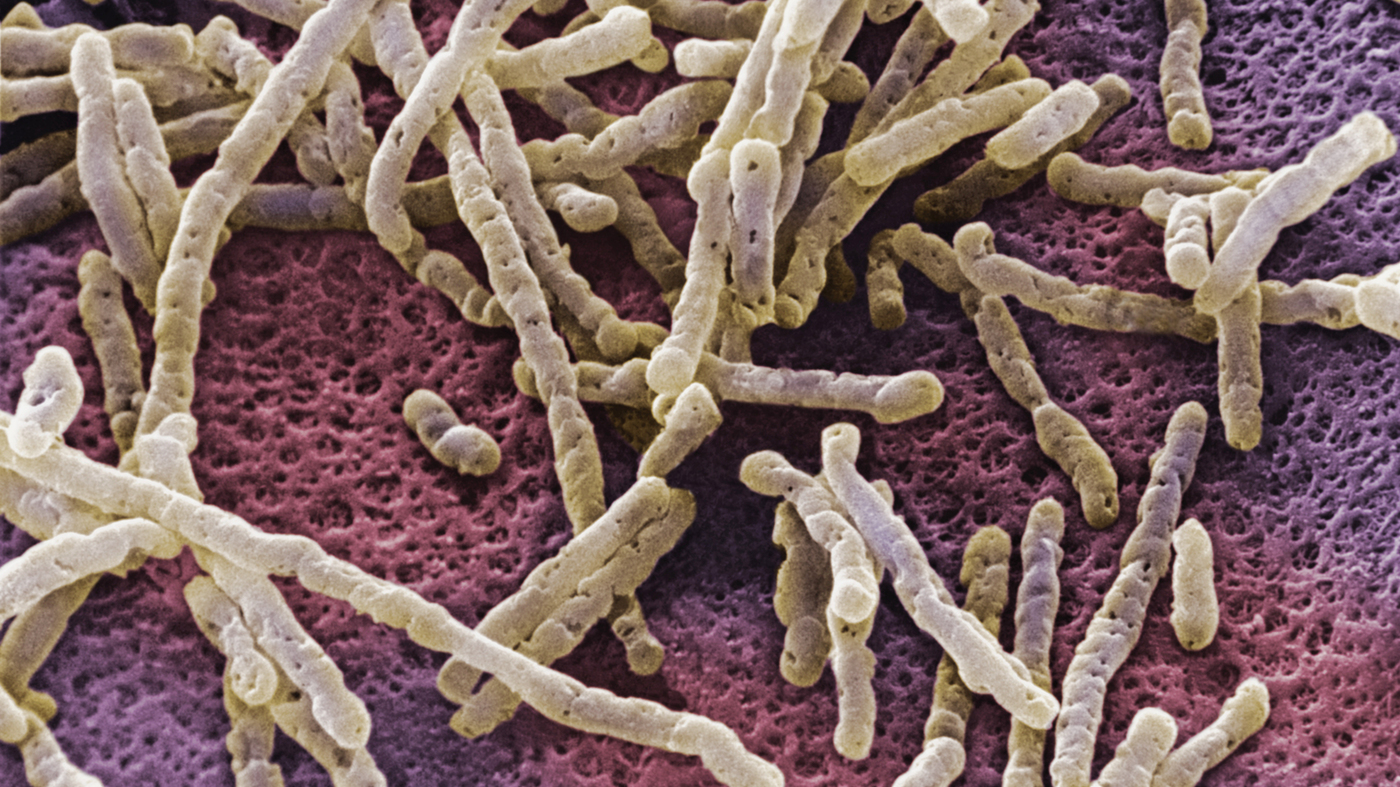
Bacterium Clostridium difficile
It turned out that Clostridium difficile has a secret weapon. One of the characteristic features of C. difficile among other intestinal bacteria is its ability to produce the compound paracresol (p-cresol) by tyrosine fermentation. P-cresol has a detrimental effect on the natural protective intestinal bacteria. Scientists have discovered that p-cresol selectively targets certain bacteria in the intestines and impairs their ability to grow. That has created a competitive advantage for C. difficile.
As evidence of this, the scientists investigated mutant C. difficile strains that could not produce p-cresol. Such bacteria were less able to compete with other types of intestinal microbiota, and thus less able to re-colonize the intestine after a primary infection. “Our study provides a new understanding of the effects of p-cresol production on healthy intestinal microflora and how it contributes to the survival and pathogenesis of Clostridium difficile. We have found that the main intestinal pathogen Clostridium difficile produces the bacteriostatic agent paracresol, which helps control the intestinal microbiota and provides C. difficile with the competitive advantages of growth, especially after the consumption of antibiotics. This unique symptom of a pathogen can provide a new drug target to reduce infections,
NLRP12 protein and Lachnospiraceae protect against obesity and insulin resistance
In another study related to intestinal microflora, scientists described the anti-inflammatory function of the protein NLRP12, which had a positive effect on the prevention of obesity and diabetes. NLRP12 promoted the growth of colonies of “good” bacteria found in the intestines, Lachnospiraceae, which produce small butyrate and propionate molecules. These substances, in turn, have a positive effect on intestinal health and prevent the development of obesity and insulin resistance.
In humans, NLRP12 is produced by several types of immune cells and appears to function as a brake against excessive inflammation. Earlier work has shown that mice with knockout of the Nlrp12 gene are very susceptible to inflammation, including experimental inflammation of the large intestine (colitis) and associated colon cancer.
In this study, scientists fed the knockout mice Nlrp12 (Nlrp12 -knockout) and normal mice with high-fat foods for several months. Nlrp12-knockout mice ate and drank no more than their healthy counterparts, but gained much more fat and gained a lot of weight. Signs of insulin resistance were also found in knockout mice, which is associated with a reduced ability to absorb glucose and, as a rule, follows the development of obesity.
The absence of Nlrp12 in these mice led to an increase in signs of inflammation in the intestines and fat deposits, but it was unclear how this resulted in an increase in excess weight. At the next stage, Nlrp12 knockout mice were divided into two groups, one of which was treated with antibiotics. As a result, weight loss was observed in mice from this group, and this allowed scientists to assume that intestinal bacteria are involved in the development of obesity. Further tests showed that when Nlrp12 knockout mice were maintained under conditions without bacteria, the mice did not gain weight, and the absence of Nlrp12 was not affected. This showed, according to scientists, that "bad" bacteria caused excessive weight gain during a diet high in fat. It is noteworthy that knockout mice were also protected from obesity when they lived together with control mice,
The researchers also injected Nlrp12 knockout mice with the beneficial bacteria Lachnospiraceae and found that these bacteria reduced intestinal inflammation, eliminated the hegemony of the harmful bacteria Erysipelotrichaceae, and increased the diversity of the microbiota. Lachnospiraceae also significantly protected animals from obesity and related kinsulin resistance. Lachnospiraceae contain enzymes that convert carbohydrates and fibers into small molecules called short-chain fatty acids (SCFA). Scientists noticed that two of them, in particular, butyrate and propionate, turned out to be much larger when the amount of Lachnospiraceae in the intestine increased. Butyrate and propionate are known to have anti-inflammatory properties that promote intestinal health.
“All of the inflammatory and metabolic changes that we observed in Nlrp12 -knockout mice during a high-fat diet were substantially canceled when we resubmitted them to Lachnospiraceae. Obesity depends on inflammation, not only on overeating and lack of exercise, and this study suggests that "good" bacteria in the intestines that help maintain a healthy weight contribute to a reduction in inflammation. In mice, NLRP12 reduces inflammation in the intestines and adipose tissues. It is also known that in obese people, the levels of NLRP12 expression are reduced, ”the researchers write [7].
Found a molecule that has potential in the prevention of Parkinson's disease
In September of this year, an article by Spanish scientists published a new molecule in the journal PNAS, which may help prevent neurodegeneration. Using a high-throughput screening analysis recently developed by them, researchers identified SynuClean-D, a small compound that inhibits α-synuclein aggregation, destroys mature amyloid fibrils, prevents the spread of fibrils, and cancels the degeneration of dopaminergic neurons in the animal model of Parkinson's disease.
Molecular events that cause the development of Parkinson's disease, lead to abnormal aggregation of α-synuclein protein in dopaminergic neurons. Α-Syn aggregation is also found in oligodendrocytes in patients with multiple systemic atrophy (MSA). α-Syn is a protein that is expressed in significant amounts in the brain. It is assumed that the α-Syn function is associated with the movement of vesicles. This protein is the main component of Taurus and Levy's neurites in Parkinson's disease.
A methodology has been developed for detecting SynuClean-D, allowing indexation of α-synuclein aggregation among thousands of different molecules. After identification, scientists conducted a biophysical in vitro test of the inhibitory activity of SynuClean-D, tests were carried out to determine the mechanism of action of SynuClean-D in human neural cell culture, before testing this substance in animal models. As such a model, two types of Caenorhabditis elegans worms were taken. They express α-synuclein in muscles and in dopaminergic neurons. Experiments have shown that the administration of the identified inhibitor, SynuClean-D, reduced protein aggregation, improved the mobility of the animal and protected it from neural degeneration.
“Everything seems to indicate that the molecule we identified, SynuClean-D, may provide therapeutic applications for the treatment of neurodegenerative pathologies, such as Parkinson's disease in the future. In general, SynuClean-D seems to be a promising molecule for therapeutic intervention in Parkinson's disease, ”the scientists write. [eight].
β-hydroxybutyrate slows down the aging of blood vessels
In September, an article by American scientists devoted to the prevention of age-related cardiovascular pathologies was published in the journal Molecular Cell. The molecule that is produced during fasting or caloric restriction has an anti-aging effect on the vascular system, which can reduce the occurrence and severity of human diseases associated with blood vessels, according to a study by the University of Georgia.
As people grow older, they are more susceptible to diseases such as cancer, cardiovascular disease or Alzheimer's disease. Age is the most important risk factor for human disease. The most important part of aging is vascular aging. With age, the vessels become very sensitive to various types of damage, so the study of vascular aging is very important.
In this paper, scientists investigated a little-known mechanism of the positive effect of calorie restriction (reducing food or fasting) on age-related processes. Researchers have identified an important small molecule that is produced during stress and under nutritional deficiencies - β-hydroxybutyrate. This substance is one of the types of ketone bodies, and is produced by the liver from fatty acids during periods of low food intake, carbohydrate-restricting diets, fasting and prolonged intensive physical exercise.
The researchers traced the entire chain of positive effects of β-hydroxybutyrate. Initially, he acted on nuclear ribonucleoprotein A1 (hnRNP A1) as a direct binding target. At the next stage, hnRNP A1 enhanced the expression of the transcription factor Oct4. And then Oct4 increased the levels of Lamin B1, a key factor opposing age-related DNA damage. Fasting and intraperitoneal injection of β-hydroxybutyrate activated Oct4 and Lamin B1 in vascular smooth muscle and in endothelial cells in mice in vivo. Scientists concluded that β-hydroxybutyrate has an anti-aging effect in vascular cells by activating the Lamin B1 pathway by inducing hnRNP A1 and Oct4. [9]
Very low and excessive alcohol consumption increases the risk of dementia.
In August 2018, an article by French and English researchers was published on the effect of different doses of alcohol on the development of dementia. 9087 participants aged 35–55 years who were monitored for 23 years as part of the Whitehall II study, which examined the influence of social, behavioral and biological factors on long-term health, were divided into three groups. The first group of participants consisted of virtually no-drinkers (scientists attributed them to the category of absistence, i.e. abstinence). The second group of participants consumed from 1 to 14 standard alcoholic units (unit) per week. Alcohol unit on average is about 12 grams of pure alcohol. In terms of alcoholic beverages, the second, moderately drinking group, it turns out, used a bottle of beer or a glass of wine a day.
As shown by the results of long-term observations, the people most at risk of dementia were people who either did not drink alcohol at all (category of abstinence) or drank more than 14 units per week. If the connection with dementia with the drinking group is quite logical and explicable, then falling into the risk category for the development of the neuropathologies of non-drinking people was a surprise for scientists.
The analysis showed that the excess risk of dementia associated with abstinence from alcohol in adulthood was partly due to cardiometabolic diseases, which were a co-factor in non-drinkers. The authors emphasize that their results should not be viewed as a call for non-drinkers to start drinking alcohol: “Our results reinforce the evidence that excessive alcohol consumption is a risk factor for dementia and encourage the use of lower alcohol consumption thresholds in guidelines cognitive health in old age. And our findings should not motivate people who do not drink to start drinking, given the known negative effects of drinking on mortality, neuropsychiatric disorders,
At the same time, in August of the same year another article was published, which described that, most likely, there is no safe dose of alcohol. As part of a large-scale study of Global Burden of Diseases, Injuries, and Risk Factors Study 2016, scientists analyzed data contained in 694 scientific papers on alcohol consumption at individual and population levels, as well as 592 prospective and retrospective studies of the risk of alcohol consumption. The data obtained showed that for health promotion it is better not to drink at all: "The level of alcohol consumption, which minimized harm to health, was zero." [2]
Prepared by: Alexey Rzheshevsky.
Bibliography
- Séverine Sabia, Aurore Fayosse, Julien Dumurgier, Aline Dugravot, Tasnime Akbaraly, Annie Britton, Mika Kivimäki, Archana Singh-Manoux. Alcohol consumption and risk of dementia: 23 year follow-up of Whitehall II cohort study. BMJ, 2018;
- GBD 2016 Alcohol Collaborators. Alcohol use and burden for 195 countries and territories, 1990-2016: Lancet. 2018 Sep 22; 392 (10152): 1015-1035.
- Mitchell SJ, Bernier M et al ... Daily Fasting Improvements for Women Composition and Calories. Cell Metab. 2018 Aug 24. pii: S1550-4131 (18) 30512-6.
- Katherine E. Paterson, Phyo K. Myint Amy Jennings, Lucy KM Bain, Marleen AH Lentjes, Kay-Tee Khaw, and Ailsa A. Welch. Vitaling Cardiovascular Disease Risk Profiles. Stroke, September 20, 2018.
- Sudharsan Periyasamy-Thandavan, John Burke, Bharati Mendhe, Galina Kondrikova, Ravindra Kolhe, Monte Hunter, Carlos M Isales, Mark W Hamrick, William D Hill, Sadanand Fulzele. MicroRNA-141-3p negatively modulates the pathophysiology of human and murine bone marrow stromal cells. The Journals of Gerontology: Series A, 2018
- Ian J. Passmore, Marine PM Letertre, Mark D. Preston, Irene Bianconi, Mark A. Harrison, Fauzy Nasher, Harparkash Kaur, Huynh A. Hong, Simon D. Baines, Simon M. Cutting, Jonathan R. Swann, Brendan W Wren, Lisa F. Dawson. Para-cresol production by Clostridium difficile affects microbial diversity and Gram-negative bacteria. PLOS Pathogens, 2018; 14 (9): e1007191
- Truax AD, Chen L et al. The Inhibitory Innate Gl Microbiota Homeostasis. Cell Host Microbe. 2018 Sep 12; 24 (3): 364-378.e6.
- Jordi Pujols, Samuel Peña-Díaz, Diana F. Lázaro, Francesca Peccati, Francisca Pinheiro, Danilo González, Anita Carija, Susanna Navarro, María Conde-Giménez, Jesús García, Salvador Guardiola, Ernest Pearls, Apes-Giménez, Jesús García, Salvador Guardiola, Ernest Apes-Giménez; , Tiago Fleming Outeiro, Esther Dalfó, Salvador Ventura. Small molecule inhibits α-synuclein aggregation, disrupts amyloid fibrils, and prevents degeneration of dopaminergic neurons. Proc Natl Acad Sci US A. 2018 Sep 24. pii: 201804198.
- Han YM, Bedarida T, Ding Y, Somba BK, Lu Q, Wang Q, Song P, Zou MH. β-Hydroxybutyrate Prevents Vascular Senescence through A1-Mediated Upregulation of Oct4. Mol cell. 2018 Sep 20; 71 (6): 1064-1078.e5.
