Genetics and chickens: human CSF1-Fc protein in egg white

Once upon a time there lived a grandfather and a woman. And they had a chicken Ryaba. The chicken laid an egg, but not simple, but genetically modified. An unexpected turn, right? But this is exactly what will be discussed today. Pharmaceuticals have changed a lot over the last dozens or even hundreds of years. We no longer use leeches and do not consider a lobotomy an excellent remedy for migraine. Now there is a great variety of drugs that can cure a variety of diseases. The basis of these drugs naturally lie certain chemical compounds, the extraction of which is sometimes associated with great difficulties. And here scientists came up with a brilliant idea - why do we need all these complex biogenerators, if we can use chickens. Yes, yes, you heard right, namely chickens that will carry eggs containing the necessary chemical compounds, such as proteins. How scientists changed chickens to achieve these goals, what came of it and whether we should not be afraid of the capture of the planet by two-meter carnivorous genetically modified hens - we learn from the report of the research group. Go.
The basis of the study However, according to scientists, the use of large animals for such purposes is quite expensive, given the small number of offspring, a long period of gestation, and then maturation, etc. That is why they turned their attention to chickens, and also due to the fact that chicken eggs contain sufficient amount of protein. The average chicken egg weighing 60 grams contains about 3.5 grams of protein. For the year, one hen can carry about 300 eggs. Add here cheap chickens and a rapid increase in numbers under normal conditions.
It should start with the fact that the use of animals as bioreactors is not something revolutionary new. For example, Antithrombin III, a preparation derived from milk of genetically modified (GM) goats, was approved for use back in 2006. In other words, the goats acted as a "laboratory" that produced milk, which included the chemical compound used in the preparation.
Also, in addition to the economic advantages, scientists note more scientific. So, in chickens and humans, glycosylation patterns, the process of attaching sugars to organic molecules, are very close. And this means that there will be a low immunogenicity of egg protein preparations that need glycosylation for activation.
All this sounds very, very interesting, but we know that chicken eggs are used more likely for an omelet, rather than as a source of medicinal bases. That is, the chickens in this study are not simple, (no, not gold, although from a financial point of view it may be so), but genetically modified.
Scientists note that lentiviruses were previously used to carry out modifications of chickens' genes, since they carry genetic information perfectly into the host cell. Transgene expression can be limited in the oviduct using the ovalbumin gene sequence (egg white albumin). This method allowed the creation of two biologically active proteins in the egg white, however, the expression level was significantly less than 1 g / l. And it is not enough for such a technique to be attractive from a commercial and production point of view.
In this study, the scientists decided to implement a more advanced method of producing two new proteins, including cytokine dimer, as well as an improved purification method. Interferon α2a, a patented biological drug for the treatment of certain forms of cancer and hepatitis (already outdated), was used as a model.
The main participant in the study was the macrophage colony-stimulating factor (CSF1), which is a 4-spiral bundle, that is, it is structurally close to many cytokines, including insulin, somatotropin, placental lactogen, etc. But the main "producer" in the study are the chickens. What results have shown experiments we consider below.
The results of the study

Image number 1
0.5 kbp (thousands of nucleotide pairs / thousands of paired bases) of the interferon α2a coding sequence were synthesized and cloned into the lentiviral vector of the replication-defective equine infectious anemia virus with EREOVA promoter.
To increase the degree of expression, 0.9 mbps of the ovalbumin promoter sequence was added between the estrogen response element and the steroid dependent regulatory element ( 1a ). It is this lentiviral vector that made it possible to obtain transgenic birds, among which a male (G1) was identified, which is the carrier of the intact (whole / intact) transgene. Accordingly, it is from this male that the line of transgenic birds (offspring) went.
Through the use of antibodies that respond to human interferon α, protein immunoblot was used to detect the presence of interferon α2a in the egg protein of test chickens ( 1b ).
Protein immunoblot * is an analytical method for determining the presence of proteins in a sample.And quantitatively α2a was described using enzyme immunoassay, when antibodies bind to specific antigens, which allows to determine their number.
To confirm the biological activity before the purification process, scientists placed a diluted egg white on a cell transfector with a reporter gene of an interferon-stimulated response element that controls the expression of luciferase ( 1c ).
Reporter gene * - joins regulatory sequences of other genes to study gene expression in cell cultures.
Luciferase * - oxidative enzymes that trigger the reaction, one of the features of which is bioluminescence.After all the checks, the cleaning process was started, which consisted of:
- clarifying ovomucin (glycoprotein in egg white) by lowering the pH;
- removal of ovomucin using a centrifuge;
- purification of interferon α2a using HiTrap Blue capture and further size exclusion chromatography.
The degree of purification (> 95%) was confirmed by polyacrylamide gel electrophoresis ( 1d ).
From 100 ml of egg protein, 15 mg of interferon α2a was obtained, and this amounts to about 60% of the volume of interferon α2a (25 mg) detected by the ELISA assay ( 1e ).
You may have a question - what about diseases that can jump from chickens to humans using this interferon α2a. Scientists have tested this, more precisely the antiviral properties of α2a by injecting H1N1 type A virus into cells previously treated with α2a. And the results of this analysis were more than positive ( 1f ) - the antiviral activity of interferon from egg white was 1x10 9U / mg, which is an order of magnitude higher than that produced by bacteria.

Image No. 2
One of the most important proteins in the differentiation, proliferation and functioning of macrophages is CSF1.
The pig CSF1 coding sequence, combined with F c , was synthesized (pCSF1-F c ) and cloned into the lentiviral pLenti6 vector (isolated from HIV) between the EREOVA2 promoter and the post transcriptional regulatory woodchuck ( 2a ) hepatitis virus .
F c * (an immunoglobulin fragment that crystallizes) is part of an immunoglobulin molecule that interacts with the F c receptor on the cell surface and with some proteins.After breeding a male (G0), which became the basis of a new line of offspring, among which an intact transgene was found in two females and one male. These individuals later took part in the breeding of the next generation (G2). Electrophoresis of proteins in a polyacrylamide gel confirmed the presence of pCSF1-F c in egg white ( 2b ).
Pig CSF1 was expressed in Ba / F3 cells. This led to the fact that the very existence of these cells was dependent on CSF1-F c . Thus, it was possible to assess the viability of the cells of the test egg protein, which was quite high in comparison with the control group (protein from normal eggs), where the survival rate was almost zero ( 2c ).
Next, egg protein from pCSF1-Fc eggs were cleared of ovomucin and transferred to the supernatant state (supernatant, liquid above dry residue), as shown in image 2d .
The pCSF1-F c active dimeric form was purified by capturing MabSelect SuRe and further polishing with size exclusion chromatography ( 2e ). The purity rate was more than 97%.
The average molecular weight was as a result of the order of 95.1 kDa (kilodalton), the monodispersity of the protein was also satisfactory - Mw / Mn = 1.011 (Mw is the weight average molecular weight, Mn is the number average molecular weight). An analysis of the copolymer model showed the presence of 85.6 kDa protein and 9.5 kDa of oligosaccharide.
Cell viability was also tested - some eggs were placed in a refrigerated chamber at -20 ° C for 1 month, and some for the same period, but at -80 ° C. In both cases, the cells survived ( 2g ), which indicates the possibility of long-term storage and their further use even before cleaning without loss of activity.
The ability of the purified protein to interact with the CSF1 receptor was also analyzed. This was done using mouse bone marrow cells, and the results were satisfactory ( 2h ).
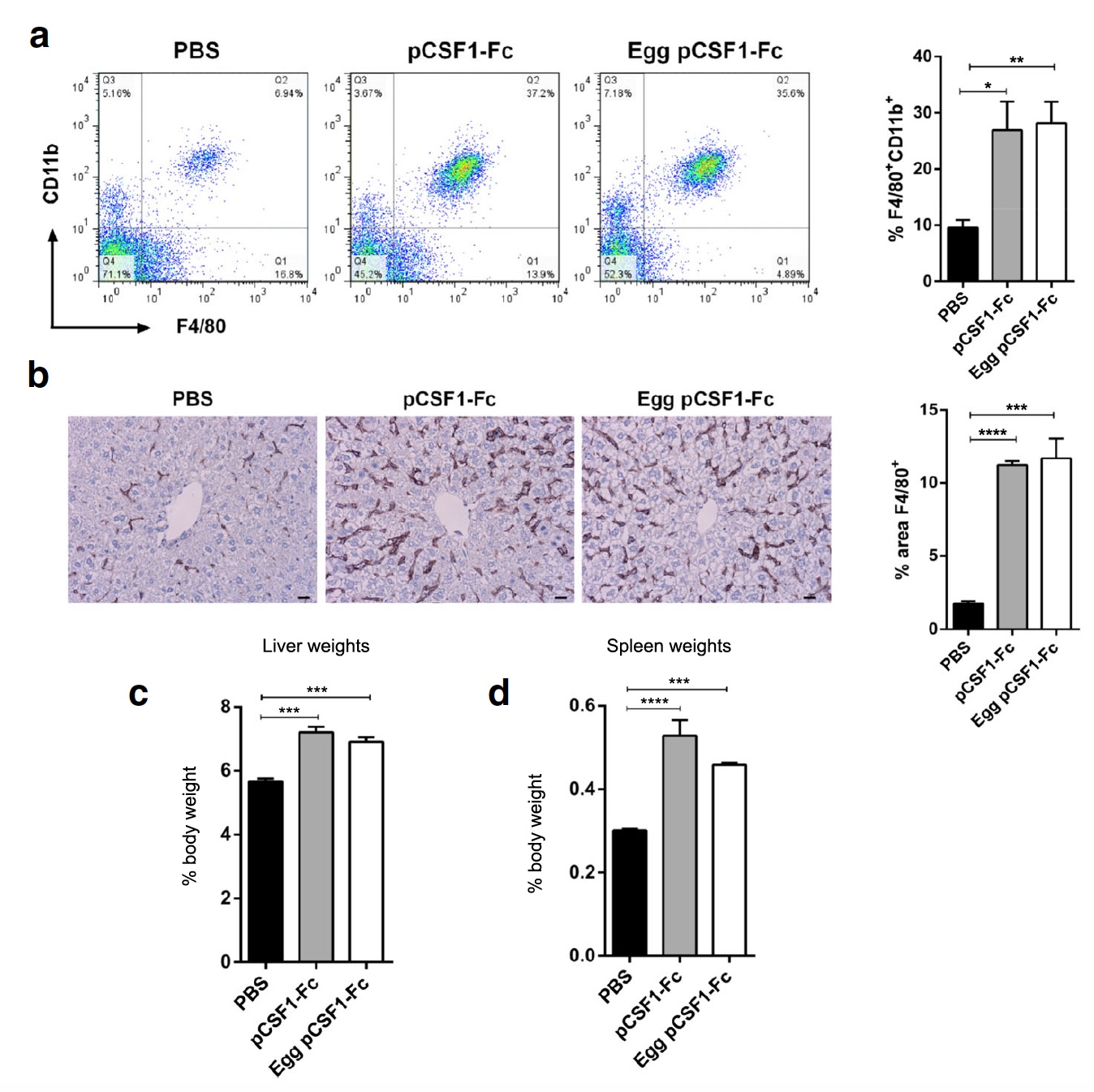
Image No. 3
Next, the scientists decided to test the activity of pCSF1-F c from egg white under field conditions, that is, on laboratory rats, which were injected with 1 μg / g of control or purified pCSF1-F proteinc . After that, the weight and histology of the liver, spleen and blood of rats were checked. Both pCSF1-F c resulted in an increase in the number of F4 / 80 + CD11b + cells in the blood ( 3a ) and macrophages of the F4 / 80 + cells in the liver ( 3b ). A significant increase in liver and spleen weight was also observed ( 3c and 3d ).

Image No. 4
The above tests, tests and experiments showed very good results. Now it remains only to prove that such methods can allow the production of human cytokines, which can be used in medicines.
Scientists note that pCSF1-F cIt shows good activity in human cells and at the same time may be immunogenic. To confirm this, laboratory rats were immunized with CSF1-F c protein to produce monoclonal antibodies that cross-react with human CSF1.
Before conducting actual tests and analyzing their results, scientists prepared chickens expressing human CSF1-F c (hCSF1-F c ). For this purpose, the methodology and vector system described earlier were used. An intact transgene was detected in two chickens, which exhibited the necessary protein expression of 1 mg / ml ( 4a ). This was followed by the above cleaning process ( 4b ). The degree of survival of purified hCSF1-F cwas also high, even after vacuum drying and reconstitution ( 4c ).
For more detailed acquaintance with the nuances of the study I strongly recommend to look into the report of scientists .
Epilogue
The result of this study is the confirmation that interferon α2a and CSF1-F c are perfectly synthesized as components of normal egg white. Extraction of these proteins also does not require much effort, but only standard methods based on chromatography. The resulting proteins are active both in the laboratory and in the body of a living test subject (rats).
Many are very skeptical of genetic modifications. Some even fear this phrase and everything that can be associated with it. However, it is worth only a little to figure out what perspectives gene modifications can give to humanity, how fears and outdated dogmas disappear. In this particular case, we looked at a study in which scientists literally forced chickens to lay eggs with human proteins, which can be used to produce cheap, effective, and fast components needed to create drugs. Maybe these chickens do not carry golden eggs, but their value is much higher.
And, of course, Friday offtop:
Немного музыки перед выходными :)
Благодарю за внимание, оставайтесь любопытствующими и отличных всем выходных, ребята.
Немного музыки перед выходными :)
Благодарю за внимание, оставайтесь любопытствующими и отличных всем выходных, ребята.
Thank you for staying with us. Do you like our articles? Want to see more interesting materials? Support us by placing an order or recommending to friends, 30% discount for Habr users on a unique analogue of the entry-level servers that we invented for you: The whole truth about VPS (KVM) E5-2650 v4 (6 Cores) 10GB DDR4 240GB SSD 1Gbps from $ 20 or how to share the server? (Options are available with RAID1 and RAID10, up to 24 cores and up to 40GB DDR4).
VPS (KVM) E5-2650 v4 (6 Cores) 10GB DDR4 240GB SSD 1Gbps until spring for free if you pay for a period of six months, you can order here .
Dell R730xd 2 times cheaper? Only we have 2 x Intel Dodeca-Core Xeon E5-2650v4 128GB DDR4 6x480GB SSD 1Gbps 100 TV from $ 249in the Netherlands and the USA! Read about How to build an infrastructure building. class c using servers Dell R730xd E5-2650 v4 worth 9000 euros for a penny?
