Ask Ethan: What are the quantum causes of the reaction of sodium with water?
- Transfer

If you put a piece of sodium in water, you can cause a violent, often explosive reaction.
Sometimes we learn something at the beginning of life and just take it for granted that the world works that way. For example, if you throw a piece of pure sodium into water, you can get a legendary explosive reaction. As soon as a piece gets wet, the reaction makes it hiss and warm up, it jumps on the surface of the water and even gives out tongues of flame. This, of course, is just chemistry. But is something else happening at the fundamental level? This is what our reader, Semyon Stopkin, from Russia wants to know:
What forces control chemical reactions, and what happens at the quantum level? In particular, what happens when water interacts with sodium?
The reaction of sodium with water is a classic, and it has a deep explanation. Let's start by studying the progress of the reaction.
The first thing you need to know about sodium - at the atomic level, it has only one proton and one electron more than that of an inert or noble gas, neon. Inert gases do not react with anything, and all because all of their atomic orbitals are completely filled with electrons. This super-stable configuration collapses when you go one element further in the periodic table, and this happens with all the elements exhibiting similar behavior. Helium is superstable, and lithium is extremely chemically active. Neon is stable, and sodium is active. Argon, krypton and xenon are stable, but potassium, rubidium and cesium are active.
The reason is an additional electron.

The periodic table is sorted by periods and groups according to the number of free and occupied valence electrons - and this is the first factor in determining the chemical properties of an element
When we study atoms, we get used to considering the nucleus as a solid, small, positively charged center, and electrons as negatively charged points in the orbit around it. But in quantum physics, this does not end there. Electrons can behave like dots, especially if you shoot them with another high-energy particle or photon, but if you leave them alone, they blur and behave like waves. These waves are capable of self-tuning in a certain way: spherically (for s-orbitals containing 2 electrons), perpendicularly (for p-orbitals containing 6 electrons), and then to d-orbitals (10 electrons each), f-orbitals ( by 14), etc.
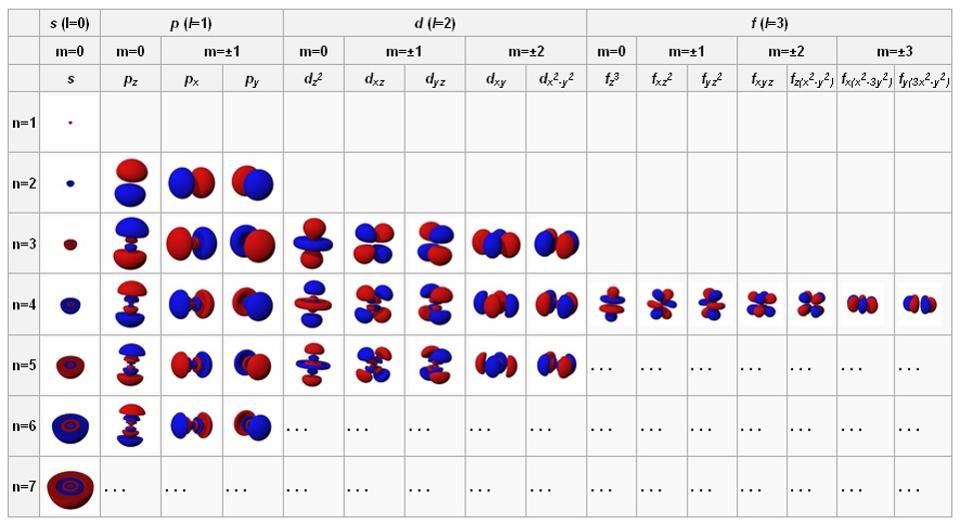
The orbitals of atoms in the state with the lowest energy are in the upper left, and when moving to the right and down, the energies increase. These fundamental configurations control the behavior of atoms and intra-atomic interactions.
These shells are filled due to the Pauli ban principle , which forbids two identical fermions(for example, electrons) occupy the same quantum state. If an electronic orbital is filled in an atom, then the only place where you can place an electron is the next, higher orbital. The chlorine atom will gladly accept an additional electron, since it only needs one to fill the electron shell. And vice versa, the sodium atom will gladly give up its last electron, since it has an extra one, and all the rest filled the shells. Therefore, sodium chlorine is so good and turns out: sodium gives an electron to chlorine, and both atoms are in the energy preferred configuration.

Elements of the first group of the periodic table, especially lithium, sodium, potassium, rubidium, etc. lose their first electron much easier than everyone else
In fact, the amount of energy needed for an atom to give up its external electron, or ionization energy, is especially low for metals with one valence electron. From the numbers it can be seen that it is much easier to take an electron from lithium, sodium, potassium, rubidium, cesium, etc., than any other element.

Frame from an animation showing the dynamic interaction of water molecules. Individual H 2 O molecules are V-shaped and consist of two hydrogen atoms (white) connected to an oxygen atom (red). Adjacent H 2 O molecules react briefly with each other via hydrogen bonds (blue and white ovals)
So what happens in the presence of water? You can imagine water molecules as extremely stable - H 2O, two hydrogen bonded to one oxygen. But the water molecule is extremely polar - that is, on one side of the H 2 O molecule (on the side opposite to the two hydrogens) the charge is negative, and on the opposite - positive. This effect is sufficient for some water molecules - of the order of one in several millions - to decay into two ions - one proton (H + ) and a hydroxyl ion (OH - ).

In the presence of a large number of extremely polar water molecules, one of several million molecules will decompose into hydroxyl ions and free protons - this process is called autoprotolysis
The consequences of this are quite important for things like acids and bases, for the processes of dissolution of salts and activation of chemical reactions, etc. But we are interested in what happens when sodium is added. Sodium - this neutral atom with one poorly held external electron - enters the water. And these are not just neutral H 2 O molecules , they are hydroxyl ions and individual protons. The protons are important to us first of all - they lead us to the key question:
What is energetically preferable? To have a neutral sodium atom Na with a separate proton H +, or a sodium ion that has lost an Na + electron together with a neutral hydrogen atom H?
The answer is simple: in any case, the electron will jump from the sodium atom to the first individual counter-proton that comes in its way.

Having lost an electron, the sodium ion will gladly dissolve in water, as does the chlorine ion, having acquired an electron. It is much more beneficial energetically - in the case of sodium - that an electron mate with a hydrogen ion.
That is why the reaction occurs so quickly and with such an energy output. But that is not all. We have obtained neutral hydrogen atoms, and, unlike sodium, they do not line up in a block of individual atoms bonded together. Hydrogen is a gas, and it goes into an even more energetically preferable state: it forms a neutral hydrogen molecule H 2. And as a result, a lot of free energy is formed, which goes into heating the surrounding molecules, neutral hydrogen in the form of gas, which leaves the liquid solution into the atmosphere containing neutral oxygen O 2 .

A remote camera shoots near the shuttle's main engine during a test run at the John Stennis Space Center. Hydrogen is the preferred fuel for rockets due to its low molecular weight and excess oxygen in the atmosphere with which it can react
If you accumulate enough energy, hydrogen and oxygen will also react! This violent burning produces water vapor and a huge amount of energy. Therefore, when a piece of sodium (or any element of their first group of the periodic table) gets into the water, an explosive release of energy occurs. All this is due to the transfer of electrons controlled by the quantum laws of the Universe, and the electromagnetic properties of charged particles that make up atoms and ions.

The energy levels and wave functions of electrons corresponding to different states of the hydrogen atom - although almost the same configurations are inherent in all atoms. Energy levels are quantized in multiples of the Planck constant, but even the minimum energy, ground state, has two possible configurations depending on the ratio of the spins of the electron and proton
So, let’s repeat what happens when a piece of sodium falls into water:
- sodium immediately releases an external electron into water,
- where it is absorbed by a hydrogen ion and forms neutral hydrogen,
- this reaction releases a large amount of energy, and heats the surrounding molecules,
- neutral hydrogen turns into molecular hydrogen gas and rises from the liquid,
- and finally, with a sufficient amount of energy, atmospheric oxygen enters into a combustion reaction with hydrogen gas.

Sodium metal
All this can be simply and elegantly explained by the rules of chemistry, and that is how it often do. However, the rules governing the behavior of all chemical reactions come from even more fundamental laws: the laws of quantum physics (such as the Pauli ban principle, which controls the behavior of electrons in atoms) and electromagnetism (which controls the interaction of charged particles). Without these laws and forces there will be no chemistry! And thanks to them, every time you drop sodium into water, you know what to expect. If you still do not understand - you need to wear protection, do not take sodium with your hands and move away when the reaction begins!
Ethan Siegel - astrophysicist, science popularizer, author of the Starts With A Bang! Wrote Beyond the Galaxy books [ Beyond The Galaxy], and Treknologiya: the science of Star Trek [ Treknology ].
FAQ: if the Universe is expanding, why aren’t we expanding ; why the age of the Universe does not coincide with the radius of its observed part .
